Bessemer Steel
Description
This section is from the book "Modern Shop Practice", by Howard Monroe Raymond. Also available from Amazon: Modern Shop Practice.
Bessemer Steel
History
During the fifties of the last century, Kelly, in the United States, and Bessemer, in England, both discovered that the carbon could be burned out of pig iron simply by blowing air through the molten metal. The Englishman was very fortunate in his aggressiveness and the prevailing conditions, and in a few years was able to develop his process so as to make a very good grade of steel immensely cheaper than it ever had been done before. Bessemer developed the furnace, almost as it is used today, and the process has been continued with comparatively little change since the time of his early successes.
Converter
All converters have nearly the same shape and are operated in about the same way. Fig. 23 shows a round body with detachable bottom. The outer casing is of heavy steel plate. It is filled and emptied through the nose A. The current of cold air enters through the pipe C, and passes through the trunnion T. It enters the converter from the windbox B, passing through the tuyeres F. The tuyeres are of fire brick 24 to 28 inches long, and have 19 holes 5/16 inch in diameter, or 7 holes 3/8 inch in diameter. The trunnion rings N are fastened to the converter, which turns on the supports for the trunnions. The bottom is coupled on with clamps and can be removed and replaced with a fresh bottom in a few minutes.
Process. Principle
In principle, the process depends upon establishing a bath of molten pig iron in a suitable receptacle provided with apertures for blowing in air, which, when it comes into contact with the hot metal, oxidizes with avidity whatever silicon, manganese, or carbon is present, and even may attack the iron itself, if the blowing is not discontinued just as the carbon is burned out. The metal thus obtained is alloyed with exactly the right amount of carbon and manganese and is cast into ingots.
Variations
Variations of the process consist in using phosphorus as the internal fuel, as can be done when using high-phosphorus pig iron and a converter which is lined with a basic instead of a siliceous refractory. By very careful work, and as practiced in some countries, the process can be stopped at exactly the right carbon content, without having first to burn out all the carbon and then to put back the right amount. American practice has found that the latter is the quicker method and that it gives good results. A surprising amount of heat is developed during the reaction, and the process must be regulated carefully. In using the acid process, one is unable to remove phosphorus and sulphur from the metal.
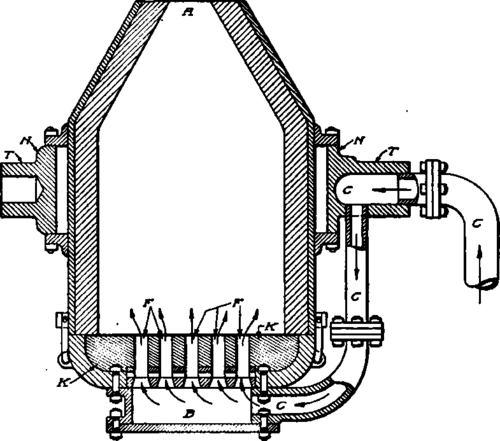
Fig. 23. Section of Round Body Converter with Detachable Bottom.
This gradually has narrowed American practice down to the use of pig iron or what is known as Bessemer grade, and the process has suffered much in comparison with the development of the open-hearth process, next to be described, which is slower but can be adjusted more at leisure.
Future Possibilities
As Bessemerizing is the most rapid and efficient method ever discovered for making steel out of iron, its further extensive use may have considerable future as part of a complex process in which the phosphorus in pig iron will be eliminated in the open-hearth furnace, the carbon eliminated in a converter, and the metal then considered finished - or given a further extra refining in an electric furnace which will bring the sulphur to a very low limit.
Continue to:


