Electrolysis, Water Purification, Electroplating. Part 2
Description
This section is from the book "Electricity For Boys. The "How-To-Do-It" Books", by J. S. Zerbe. Also available from Amazon: Electricity for Boys.
Electrolysis, Water Purification, Electroplating. Part 2
Composition Of Water
Now, water, in which harmful germs live, is one-third oxygen. Nevertheless, the germs thrive in water, because the oxygen is in a compound state, and, therefore, not an active agent. But if oxygen, in the form of gas, can be forced through water, it will attack the germs, and destroy them.
Common Air Not A Good Purifier
Water may be purified, to a certain extent, by forcing common air through it, and the foulest water, if run over rocks, will be purified, in a measure, because air is intermingled with it. But common air is composed of four-fifths nitrogen, and only one-fifth oxygen, and, as nitrogen is the staple article of food for bacteria, the purifying method by air is not effectual.
Pure Oxygen
When, however, oxygen is generated from water, by means of electrolysis, it is pure; hence is more active and is not tainted by a life-giving substance for germs, such as nitrogen.
The mechanism usually employed for purifying water is shown in Fig. 90.
A Water Purifier
The case (A, Fig. 90) may be made of metal or of an insulating material. If made of metal it must be insulated within with slate, glass, marble or hard rubber, as shown at B. The case is provided with exterior flanges (C, D), with upper and lower ends, and it is mounted upon a base plate (E) and affixed thereto by bolts. The upper end has a conically-formed cap (F) bolted to the flanges (C), and this has an outlet to which a pipe (G) is attached. The water inlet pipe (H) passes through the lower end of the case (A). The electrodes (I, J) are secured, vertically, within the case, separated from each other equidistant, each alternate electrode being connected up with one wire (K), and the alternate electrodes with a wire (L).
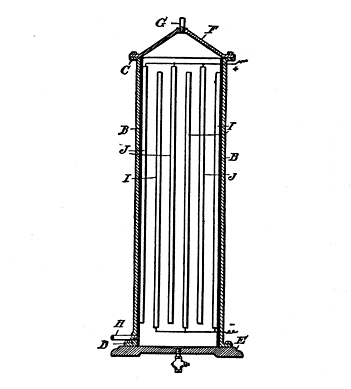 Fig. 90. Electric Water Purifier
Fig. 90. Electric Water Purifier
When the water passes upwardly, the decomposed or gaseous oxygen percolates through the water and thus attacks the germs and destroys them.
The Use Of Hydrogen In Purification
On the other hand, the hydrogen also plays an important part in purifying the water. This depends upon the material of which the electrodes are made. Aluminum is by far the best material, as it is one of nature's most active purifiers. All clay contains aluminum, in what is known as the sulphate form, and water passing through the clay of the earth thereby becomes purified, because of this element.
Aluminum Electrodes
When this material is used as the electrodes in water, hydrate of aluminum is formed, or a compound of hydrogen and oxygen with aluminum. The product of decomposition is a flocculent matter which moves upwardly through the water, giving it a milky appearance. This substance is like gelatine, so that it entangles or enmeshes the germ life and prevents it from passing through a filter.
If no filter is used, this flocculent matter, as soon as it has given off the gases, will settle to the bottom and carry with it all decomposed matter, such as germs and other organic matter attacked by the oxygen, which has become entangled in the aluminum hydrate.
Continue to:


