Gelatine Argentic-Bromide Emulsions And Plates
Description
This section is from the book "American Library Edition Of Workshop Receipts", by Ernest Spon. Also available from Amazon: American Library Edition Of Workshop Receipts.
Gelatine Argentic-Bromide Emulsions And Plates
The formulae, apparatus, and mode of working described are those which experience has found to be useful and practical. Any person, by • following these directions, may succeed in making for himself excellent rapid dry plates.
As a general rule, prepare the emulsion in the evening, and the formulae to be given should make 22 oz. of finished emulsion, with which 54 6 1/2 x 8 1/2 glass plates may be coated.
The emulsion should be mixed by a non-actinic light, say, in the form of a good-sized lantern, 6 in. square by 10 in. high, having inside a kerosene lamp with a long chimney. The front and sides are glazed with one thickness of ruby glass, the front pane being covered with one, and the side panes with two thicknesses of yellow orange post-office paper. When preparing the emulsion, or coating plates, the light is turned up only to half its full height. The lantern is fixed on a shelf 1 ft. above the table and about 3 ft. from the place of mixing or coating.
First make the following two solutions: -
No. 1.
Potassium bromide .. .. 20 grm.
Gelatine, Nelson's No. 1 .. 3 grm.
Potassium iodide .. .. .. 4 gr.
Distilled water .. .. .. 170 cc.
Alcohol, 95 per cent..... 25 cc
No. 2. Silver nitrate .. .. 25 gr. Distilled water ......170 cc.
On the following evening make as follows: -
No. 3.
Heinrichs' special gelatine ... 34 gr.
Nelson's No. 1 gelatine .. 4 gr.
Distilled water .. ., .. 180 cc.
Each of the above solutions may be prepared separately, in ordinary light. Solution No. 1 is placed in a water bath of warm water, and constantly stirred with a glass rod; when all the ingredients are completely dissolved, it is tested with blue litmus paper for acidity; if not acid, it is made so by the addition of enough of the following to turn the litmus slightly red: -
Distilled water .. .. 5 dr. Acetic acid.. .. .. 1 dr.
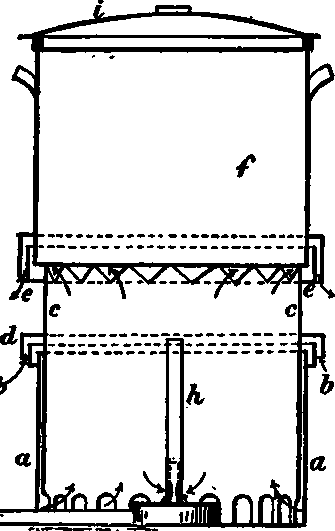
The first apparatus that is necessary is a good heating arrangement for raising the temperature of the solutions. Fig. 81 is a sectional view of the heater. The outside cylinder a is made of sheet iron, and riveted at its upper end is an upwardly projecting air-inlet flange b. Sitting inside the outside cylinder a, with its upper and lower edges escalloped or serrated, is an inside cylinder c. A downwardly projecting air-inlet flange d is riveted to the outside of cylinder, so that it projects just over the flange b on cylinder a. A second upwardly projecting inlet flange e is riveted to the upper end of cylinder. Resting on the upper edge of the cylinder c is an ordinary metal boiler /, having a downward projecting inlet flange g at the bottom, which overlaps the flange e on the cylinder c. The cover of the boiler has projecting flanges, making a light-tight joint when placed in position.
Inside the cylinder c is put a Bunsen gas burner or kerosene lamp A, a close joint being made where the supply tube passes through the outside cylinder a. It will be seen that the above construetion permits of the burning of the gas or lamp in a dark room without danger of the light escaping. All the projecting flanges must be blackened on the inside. In place of the boiler f a smaller and more shallow boiler maybe put on, to be used as a hot water bath, which will be found necessary to keep some of the solutions at a uniform temperature. The air to supply the burner h passes between flanges b and d, down between cylinders a and c, through apertures at the bottom of the latter, as shown by the arrows, while the heat escapes at the top of cylinder c, between flanges e and g.
Fig. 81.
Three quarts of cold water are put into the boiler / (Fig. 81), which should be at the boiling point by the time solutions Nos. 1 and 2 are prepared. The boiler is next removed, and in its place is put the smaller boiler or water bath. In a 10 oz. glass beaker is placed solution No. 1, and in the flask a (Fig. 82) solution No. 2. the cork and tubes having been removed. The beaker and flask are next placed in the shallow water bath, and enough cold and hot water from the boiler is poured into the bath to surround them without danger of floating or upsetting, the temperature of the water being no higher than 110° F. From the lamp underneath, the temperature of the surrounding water is gradually raised until, by a thermometer immersed in solution No. 1 in the beaker, a temperature of 155° F. is indicated.
Fig. 82.

The whole apparatus and operations should now be transferred to the dark room.
The water bath, with the beaker and flask in it, is now removed from the heater, and set upon a table, the boiler previously removed is again returned to the heater, and the water in it is left to boil, while the following is being done: -
The cork and bent glass tubes are inserted in the flask a (Fig. 82), then the No. 1 solution is poured from the beaker into a second 24 pz. flask b, and in it is inserted the pipette tube c of flask a. Holding flask 6 by the neck in the left hand (Fig. 82), the operator by blowing through the tube forces the silver solution in a fine stream into the bromide solution until the flask is emptied. The flask b is continually rotated by the left hand, while the flask 'a is steadied in a water bath by the right hand. The constant agitation of the No. 1 solution in flask b is necessary during the injection of the silver solution, in order that the silver may be completely converted into bromide. The cork and tubing are next removed from flask a, and the latter is rinsed with 20 cc. of distilled water, which is added to the mixed emulsion in the flask 6. A piece of clean cloth is tied over the mouth of the latter, and it is. then placed in the boiler, the water in which is at the boiling point. The cloth over the mouth of the flask prevents the water of condensation in the boiler from dropping into the flask.
Continue to:


