Cooling Air. Part 14
Description
This section is from the book "American Library Edition Of Workshop Receipts", by Ernest Spon. Also available from Amazon: American Library Edition Of Workshop Receipts.
Cooling Air. Part 14
Its total capacity for the work proposed, should be 27 pints of volatile liquid, and each of the 3 moulds will contain about 12 oz. of water - say, together, about 2} lb.
In Pictet's larger apparatus, the metallic vessels containing the water to be frozen are surrounded by a mixture of glycerine and water. The sulphurous oxide is drawn into a copper tubular refrigerator, the liquid filling the space between the tubes. Here takes place vaporisation, with the consequent production of intense cold, and the temperature of the non-congealable mixture of glycerine and water surrounding the refrigerator is so far reduced that water placed in the metallic vessels immersed in the tank rapidly becomes frozen. A propeller-wheel sends a current of the glycerine solution through the tubes, and thus hastens the refrigeration. The vapour of the oxide is drawn out of the refrigerator by the pump, and forced into the space between the tubes of the condenser. Through thes. tubes, a stream of cold water is constantly forced; this determines the condensation of the vapours, and the re-liquefied oxide passes into the admission-pipe, and enters again into circulation. A saturated solution of magnesium chloride gives better results than the glycerine mixture.
The tension of the oxide vapour varies from about 14.7 to 13 lb.; on the return stroke, the gas is compressed to 1/4 or 1/6 its original volume, having its temperature raised to 200°; F. (93° C). The cold water current reduces this temperature to about 61° F. (16° C.) at the outlet; and under a pressure of 3-31/2 atmos., the gas resumes a liquid state. It is claimed that 1 lb. of acid produces nearly 1 lb. of ice; and that with a consumption of 22 1/2 tons of coal, 250 tons of ice can be made every 24 hours. The cost is said not to exceed l c. a lilo. (say 1/2d. a lb.). The system is largely adopted in skating-rinks, breweries, etc.
Various - Liquid Machines
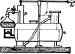
Some machines can work with any volatile liquid. One of the earliest machines of this kind, and so simple that it can be made at home, was that invented by Jacob Perkins in 1834, and shown in Fig. 22. The apparatus is carried on a wooden base a, about 5 ft. long and 2 1/2ft. wide. At one end is a jacketed copper pan 6, the interior of which holds the water to be frozen, while the jacket c contains the volatile liquid and its vapour. The pan b is enclosed in a wooden box d, containing powdered charcoal e as a non-conductor. From the top of the jacket c, a pipe f is led away to the auction-valve of an air-pump g, fixed in the middle of the wooden base a. From the delivery - valve of this pump, a pipe A proceeds to the top of a worm icontained in a worm-tub k, at the farther end of the base a. The worm - tub is supplied with water through an inlet I at the bottom, while the escape is at m. A pipe n in con-tinuation of the lower end of the worm is connected with the under side of a valve-box o, in which is a valve loaded to about 15 lb. per sq. in., so that the vapour in the worm is subjected to this pressure, as well as to the cooling influence of the water, and by these means is brought back to a liquid condition.
From the upper side of the valve-box a pipe p proceeds to the bottom of the jacketed pan, to convey the liquor to it, thus completing the circuit. The box d has a doable lid lined with charcoal, and a pane of glass at r.
Fig. 22.

Another machine adapted to the use of almost any volatile liquid, whether ammonia, carbon bisulphide, ether, be., is Holden's, of Philadelphia. It was first designed solely for reducing the temperature of liquids, such as beer, when the liquid to be cooled is allowed to trickle down over the refrigerating coil It has been widely adopted in American breweries. For the production ofice, additional plant is necessary, consisting of a large tank, and suitable receptacles for the water. A novelty in Holden's arrangement is that the waterholding vessels are introduced at one end of the tank and removed at the other, passing through progressively.
Water Machines
It has long been known that extreme cold can ha produced by the rapid evaporation of water in a comparatively perfect vacuum, the heat required for vaporisation being abstracted from the remaining water, which consequently becomes reduced in temperature, and if the process be sufficiently prolonged, actually converted into ice. Machines to carry out this principle have been constructed by Leslie, Carre, and others, but in all these cases the air-pump served only for the rarefaction of the air in the refrigerating compartment, and not for the removal and condensation of the vapour, which had to be entirely absorbed by sulphuric acid, requiring renewal after each operation. Owing to this defect, continuity of action could not be obtained, while the removal and replacement of the acid was not only an expensive operation, but was open to obvious objections from the danger and difficulty of dealing with such a highly corrosive material as oil of vitriol. For these reasons the introduction of vacuum machines has never been general, and in point of fact they were little known or used, except for producing very small quantities of ice for household purposes and for laboratory experiments, in both of which cases the air-pump was worked by hand. (Engineer.)
Windhausen, and others, who have taken up the practical improvement of his inventions, have brought to a commercial success the manufacture of ice by the evaporation of water at a very low pressure. Their machinery is now working in Berlin and at the Aylesbury Dairy Co.'s premises in St. Petersburg Place, Bayswater. The evaporation of part of the water and freezing of the remainder is effected in 6 vessels of truncated conical form, into which the water is allowed to flow at a regulated speed from 6 tanks above the cones.
Continue to:


