Distilling. Part 2
Description
This section is from the book "American Library Edition Of Workshop Receipts", by Ernest Spon. Also available from Amazon: American Library Edition Of Workshop Receipts.
Distilling. Part 2
The frames being laid on the ground, it is difficult to discover a leak, and the wood in the sides of the roof, between the fresh-water groove and the salt water, is apt to crack in the part above the level of the salt water, and cause a loss of fresh water by its leaking back into the trough. The first defect could be remedied, at a moderate cost, by raising the longitudinal timbers on cross-sleepers placed 4 or 5 ft. apart, and the second defect by lining the grooves with thin sheet-lead or tin. In the warm vapour under the glass, iron is very quickly destroyed. The temperature of the water in the troughs at noon (when the thermometer stands at 80° P. in the shade), is 140° to 150° F. The distillation usually begins at about 10 a.m. and ends at about 10 p.m.
Some experiments were made, but very incompletely, to try the effect of warming the water in a boiler before it entered the troughs, especially for use during the night and early morning. From the little that was done, it appeared probable that good results might he expected. On cloudy days, the production is less than one-half, about 40 per cent. of that on sunny days. Cloudy days are, however, very rare in the locality. (Harding.)
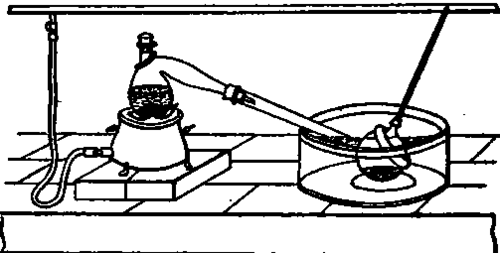
All ordinary distillating apparatus consists of 2 parts - one in which the heat is applied to the body to be distilled and vaporiaed (called the "still"), and the other into which the vapours that are formed enter in order to undergo the refrigeration that condenses them (termed the "condenser"). One of the simplest forms of distillating apparatus used in laboratories (Pig. 88) consists of a still into which is introduced the liquid to be distilled, and which is placed upon a furnace. The neck of this fits into that of a sphere whose opening must be wide enough to allow the orifice of the still to reach the spherical part of the receiver. Finally, the sphere dips into a vessel full of cold water, and is cooled on its external surface by a wet cloth. The heated mixture begins to boil, and its vapours escaping from the retort, cool and condense upon the cold sides of the spherical receiver. This latter serves at once as a refrigerator and a vessel for receiviug the distilled product.
Fig. 88.
In the beginning, the empty receiver weighs less than the volume of water that it displaces, and tends to float. It is often kept in the water by placing a weight (a brick, for example) upon the top of its spherical part and upon the sides of the vessel containing the cold water. The want of stability of this much used arrangement makes it un-recommendable. It is preferable to make use of a sufficiently heavy ring of lead into which the neck of the receiver may be introduced, and which may rest upon the latter's bulge. Upon fixing a similar ring under the receiver, the latter will be prevented from turning laterally and even from getting broken. The water in the external vessel is renewed so as to keep it cold.
A simple arrangement of this kind is not adapted for materials that have a low boiling-point, since a large proportion of the vapours escapes, and makes its exit through the neck of the receiver, which is kept hot by the vapours coming from the still. The following, which is just about as simple, is a much more perfect arrangement.
Fig. 89.
The narrow part of the still is fixed into the neck of a long, tubular receiver (Fig. 89) by means of a cork which it traverses. This annular cork exactly closes the space between the neck of the still and that of the receiver. On the other side, in the tubulure of the receiver there is fixed, by means of a cork, perforated and arranged like the preceding, a long and narrow glass tube.
When the still has been filled with the substance to be distilled, and placed upon a furnace covered with wire gauze, the receiver is immersed, as above stated, in cold water. The vapours that are formed become cooled in traversing the-elongated neck of the receiver, and are thoroughly condensed in the immersed part, provided the ebullition is not too rapid. In this latter case, the narrow tube, which presents the only open orifice, becomes heated, and indicates to the operator that the fire must be moderated.
The inconvenience of every apparatus of this kind is that the vapours which enter the receiver are not compelled to impinge against the sides, and may go directly to the exit-tube, or, in other words, the refrigeration is not methodical. Moreover, the refrigerating surface continues to diminish in measure as the receiver fills. Finally, if the receiver breaks, the entire distilled product comes in contact with the water. Despite these disadvantages, the rapidity with which such apparatus may be arranged causes them to be frequently employed.
The use of refrigerators permits of a more exact and methodical condensation of the vapours. These are arranged as follows: The 2 orifices are placed in contact by means of a rubber tube, 3 to 4 cm. in length, into one end of which is introduced the neck of the retort a (Fig. 90), and into the other the tube of the refrigerator. The latter being held in an inclined position by means of a clamp, a current of water traversing it from top to bottom, and a bent tube being adapted to its lower extremity, the free extremity of the bent one is fixed into the flask that is to collect the product. We may also suppress the central tube of the refrigerator in the flask 6, kept inclined. To facilitate this arrangement, the neck of the retort is cut at a point where it has the same external diameter as the tube of the refrigerator, and is then edged with a flame. Again, if the difference between the diameters is considerable, we may, by means of a flame, draw out slightly the one of the two tubes that is the larger, and cut it at the proper point to obtain an equality in the diameters. Finally, we may solder to the extremity of the refrigerator a cylindrical tube, 2 or 3 cm, in diameter and 6 or 7 in length, into which is fitted the neck of the retort previously provided with a cork.
Continue to:


