149. The Formation Of Alkalies
Description
This section is from the book "Applied Science For Metal Workers", by William H. Dooley. Also available from Amazon: Applied Science For Metal Workers.
149. The Formation Of Alkalies
Alkali is the commercial and industrial name for a strong base, such as caustic soda (NaOH), caustic potash (KOH), and ammonium hydroxide (NH 4OH). An alkali is opposite to an acid in character and turns red litmus paper blue. When a limited amount of acid is added to an excess of alkali, only part of the OH (hydroxide radical) is replaced by a negative element or radical, and a basic salt is formed.
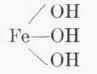
To illustrate: If excess iron (ferric) hydroxide and a limited amount of hydrochloric acid are mixed the result is:
Fe(OH)3 + HCl = FeCl (OH)2 + H 2O or
| + | HCl | = |
| + | H1O |
Ferric Hydroxide | Hydrochloric Acid | Basic Ferric Chloride |
The most important of the alkalies in caustic soda which is made from soda ash by adding milk of lime (calcium hydroxide) to the solution.
Na2CO3 | + | Ca(OH)2 | = | CaCO3 | + | 2NaOH |
Soda Ash | Milk of Lime | Calcium Carbonate | Caustic Soda |
The calcium carbonate separates as a sediment.
Continue to: