208. Battery Cells
Description
This section is from the book "Applied Science For Metal Workers", by William H. Dooley. Also available from Amazon: Applied Science For Metal Workers.
208. Battery Cells
When electricity is desired for bells, burglar alarms, etc., it is obtained from battery cells. The electricity is generated by chemical means. There are many forms, each of which has its advantages and disadvantages. The four types most commonly used are described below.
The Leclanche cell consists of a glass jar containing a solution of sal ammoniac (ammonium chloride) with a zinc rod for one pole and a carbon plate in a block of compressed manganese dioxide for the other. The purpose of the manganese dioxide is to prevent polarization of the cell, that is, the collecting of bubbles of hydrogen on the plate. Polarization diminishes the voltage by increasing the resistance. As the manganese dioxide is in a powdered form it hardens slowly, and if too large a current is taken from the cell, polarization takes place. The advantage of this type of cell lies in its freedom from local action and in the fact that it can be used for a long time without deterioration.
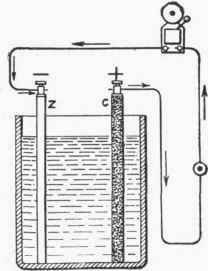
Fig. 74. - Simple Voltaic Cell.
The Daniell cell consists of a zinc sulphate solution and a copper plate in a copper sulphate solution. They are separated by a porous cup to prevent undue mixing. As no hydrogen is developed in this cell, there can be no polarization. When the circuit is left open the copper coats the zinc and impairs its efficiency.
The gravity cell consists of a copper sheet placed in the bottom of a glass. Crystals of copper sulphate are placed over the plate and water is added until the jar is nearly full. A zinc plate is suspended at the top of the jar and sulphuric acid added to start the cell. The sulphuric acid acts on the zinc forming zinc sulphate. The zinc sulphate is so much lighter than copper sulphate that, so long as the cell is kept on a closed circuit, the solution mixes but slightly.
The bichromate cell is used for operating small cells or motors, and consists of a zinc and carbon plate in a solution of chromic acid (mixture of bichromate and sulphuric acid). When the cell is not used, the zinc must be removed from the liquid, so that the chromic acid will not attack it.
Continue to:


