An Apparatus For Preparing Sulphurous, Carbonic, And Phosphoric Anhydrides
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
An Apparatus For Preparing Sulphurous, Carbonic, And Phosphoric Anhydrides
By H.N. Warren, Research Analyst.
Having had occasion to prepare a quantity of sulphurous anhydride, for the purpose of reducing chromates previous to their analysis, I made use of the following apparatus, as represented in the accompanying figure. It consists of a glass vessel, A, provided with three tubulars, otherwise resembling a large Wolff bottle, the large tube, B, being provided with a stopper for the purpose of introducing pieces of sulphur from time to time into the small dish, C, intended for its reception, and fed with air by means of the delivery tube, D, thus allowing the stream of gas caused by the consumption of the sulphur to escape by means of the exit tube, E, to the vessel desired to receive it.
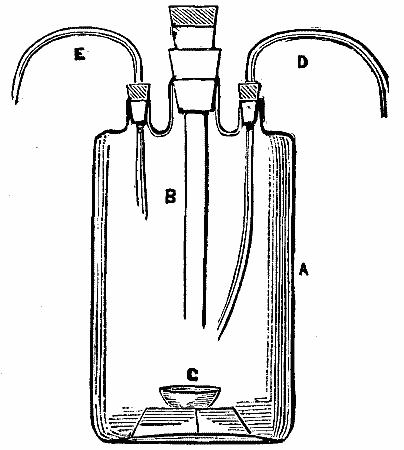
In using the apparatus the sulphur is first kindled by introducing a red hot wire through the tube, B, and replacing the stopper that has been momentarily removed for the introduction of the same. A slight blast is now maintained from the bellows that are in connection with the pipe, D, until the whole of the sulphur is thoroughly kindled, when a somewhat more powerful blast may be applied. When the apparatus above described is in full working order, from 2 to 3 lb. of sodium carbonate may be converted into sodium sulphite in less than half an hour, or several gallons of water saturated. I have also on connecting the apparatus with a powerful refrigerator obtained in a short time a large quantity of liquid SO. It will be found advantageous, however, during the preparation of sulphurous anhydride, to employ a layer of water covering the bottom of the vessel to about 1 inch in depth. Carbonic anhydride and phosphoric anhydride may also be readily obtained in any desired quantity by slight alteration; but in case of phosphorus the air must be allowed to enter only gently, since a rapid current would at all times determine the fracture of the vessel. - Chem. News.
Continue to:


