Factors Affecting The Ease Of Formation And Stability Of Mayonnaise. Continued
Description
This section is from the book "Experimental Cookery From The Chemical And Physical Standpoint", by Belle Lowe. Also available from Amazon: Experimental cookery.
Factors Affecting The Ease Of Formation And Stability Of Mayonnaise. Continued
Kilgore suggests the following method. The salt, mustard, and other dry seasonings are added at first to fresh egg yolks, for they aid in holding the excess moisture. To this is added just enough vinegar to make a heavy paste. Then run in the oil until half is added, add another portion of vinegar, equal to that at the start, and the remainder at the end of the batch.
Corran suggests that the emulsification be carried out in three stages. (1) The dry ingredients, egg yolk, and part of the aqueous ingredients are mixed thoroughly. (2) All the oil is added, forming a creamy nucleus. (3) The creamy nucleus is diluted with the remainder of the aqueous ingredients.
The ingredients used. These include the oil, the salt and seasonings, the vinegar, the egg yolks, and even previously emulsified mayonnaise. Any of the edible oils may be used in mayonnaise.
Salt and seasonings. Salt, mustard, paprika, and pepper are usually added to mayonnaise. Small amounts of other seasonings may be used. Pasteurized spice mixtures are also now available to the mayonnaise manufacturer. The addition of an electrolyte to an emulsion may produce different results, depending upon the electrolyte, the emulsifier, and the type of emulsion. Some electrolytes cause reversal of certain emulsions; others may tend to stabilize the emulsion. Seifriz in his work with emulsions stabilized with casein found that sodium chloride had no influence on the water-in-oil emulsions, but tended to stabilize oil-in-water emulsions. The effect of sodium chloride would probably vary with the emul-sifier and the concentration of the salt. Krantz and Gordon found that the concentration of the sodium chloride affected the stability of some emulsions. In mayonnaise, when egg yolk is used as the emulsifier and with the proportion of salt given in Experiment 52, it has been found that the addition of the salt to the egg yolk before the addition of any oil tends to stabilize the emulsion. In these experiments the vinegar was also added to the egg yolk before the oil was added. When the salt is added to the mayonnaise after the oil has been added, the size of the dispersed oil globules is larger and the emulsion breaks more readily while it is being made, indicating a lessened stability. The amount of salt used in the recipe may tend to lessen the solubility of the egg proteins sufficiently so that a more tenacious film is formed. If the concentration of the salt is great enough to "salt out" the egg proteins, then different results would be obtained. Thus it seems preferable to add the salt to the egg yolk and vinegar before adding the oil in making mayonnaise. Calcium salts, since they tend to form water-in-oil emulsions, are detrimental to mayonnaise. Hence it is recommended that the salt used should contain less than 0.1 per cent of foreign calcium salts. Kilgore's results indicate it is preferable to add the mustard to at least part of the vinegar to make a paste. This is then added to the egg yolk.
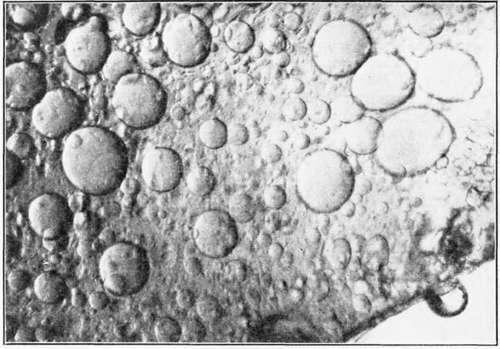
Fig. 27. - Mayonnaise. Showing the coarse emulsion formed after the addition of one tablespoon of oil. The vinegar and seasonings were added to the egg yolk, before the oil was added. Magnification approximately x 200.

Fig. 28. - Mayonnaise. Same as Fig. 27, but after adding one-fourth cup of oil. Magnification approximately x 200.

Fig. 29. - Mayonnaise. Same as Fig. 28, but after adding three-eighths cup of oil. As the oil spheres become smaller with the addition of more oil the mayonnaise becomes stiffer. Magnification approximately x 200.

Fig. 30. - Mayonnaise. Same as Fig. 29, but after the addition of one-half cup of oil. Magnification approximately x 200.
Egg yolks. Johnson and Mark have reported that cold-storage eggs are inferior to fresh eggs for making mayonnaise.
However, frozen yolks, either salted or sugared, are more extensively used at the present time than fresh yolks. Kilgore states the fresh yolk is usually too light in body to produce a good emulsion at the start. This is true in spite of the fact that fresh yolk is the best possible emulsifier and that less can be used than is required for frozen yolk. The freezing changes the physical consistency of the yolk. Upon defrosting the frozen yolks are very thick and paste like, owing to the binding of the water during freezing.
The addition of a small amount of emulsified mayonnaise. Mayonnaise is very easily made if from 1/4 to 1/3 cup of previously made mayonnaise is added to the egg yolk and vinegar. This gives a larger quantity of material to work with, but if an emulsion is already started the emulsification of additional oil is accomplished more readily. After an emulsion is once formed, subsequent additions of oil are very easily emulsified. This is analogous to seeding in crystal formation and the addition of an old gel to gelatin to hasten jelly formation. Egg yolk is itself an emulsion containing about 30 per cent of fat.
Kilgore states, "another solution to the problem of producing high consistency mayonnaise having medium or low yolk content, is to start the emulsion by means of some finished mayonnaise added to the yolk and spices. This trick of starting an emulsion, or 'seeding' it, by means of one previously made is common in pharmaceuticals. It is the fundamental principle of a process for continuous emulsification."
Temperature. Clayton states that, in general, the effect of rise in temperature is to make emulsification easier. The reason for this is that viscosity is reduced with a rise in temperature, but more important, a rise in temperature is accompanied by a decrease in the interfacial tension of non-miscible liquids. Branch states that in commercial production of mayonnaise the temperature of the mixing room is kept at 65° to 75°F.
Continue to:


