Kinds Of Sugar Used In Jelly
Description
This section is from the book "Experimental Cookery From The Chemical And Physical Standpoint", by Belle Lowe. Also available from Amazon: Experimental cookery.
Kinds Of Sugar Used In Jelly
Most jelly is made with sucrose. There are two sources of sucrose: one, the sugar beet; the other, sugar cane.
Cane or beet sugar. For some unexplained reason many persons believe that jelly cannot be made from beet sugar. Shaw in a series of over 2000 tests found no difference in the two sugars. Lowe and Redfield, working in a section of the country where many persons believe that beet sugar will not make jelly, could detect no differences in jellies made from beet and cane sugar. The sugar was sent from different sections of the state. In these experiments the source and kind of sugar were not known until the experiments were finished.
Dextrose. Crystalline dextrose when substituted by weight for sucrose in a fruit juice yields a jelly similar in texture to that obtained with sucrose. It is not so sweet as the jelly made with sucrose. In the jelly made with dextrose, crystallization of the sugar occurs. Crystals usually begin to form at the top of the surface of the jelly in 24 hours. They increase until the whole mass is crystallized. The shape of the masses of crystals resembles coral formation or reminds one of the flowerets of cauliflower except that they are colored. See Fig. 18. If the glass is full of jelly the crystals push up the top of the lid and bulge out over the side. Sometimes the first crystals appear in spots throughout the jelly instead of on the surface. Sometimes the first crystals are longer than 24 hours in appearing. If the jelly is turned out of the glass, crystallization occurs rapidly and the whole mass is usually crystallized in 48 hours or less. If dextrose is substituted for 50 per cent of sucrose, crystallization may not occur, and if crystals do appear they are slower in forming. About 50 per cent of the jellies made in the author's laboratory with one-half sucrose and one-half dextrose crystallized.
Levulose. Levulose also forms jelly, but it is more likely to be sirupy, and if a jelly is formed the sirup must be boiled to 107°C, which brings the levulose nearer to its saturation point.
Maltose. Maltose has been used by the author for gooseberry and currant jelly. Maltose is considerably less soluble than sucrose and a little less soluble than dextrose at room temperatures. The jelly made with maltose is very tart and crystallizes as readily, or more so, as jelly made with dextrose.
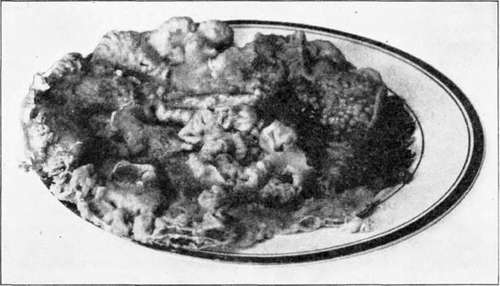
Fig. 18. - Jelly made from crystalline dextrose. Showing crystal formation 48 hours after the jelly had been turned out of the glass and 72 hours after the jelly was made.
Continue to:


