Saponification
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Saponification
Through scientific investigation it has been established that many volatile oils contain ester-like compounds, the components of which, on the one hand are alcohols, usually of the composition C10H18O or Cl0H20O and acid radicals of the fatty series, on the other.
1) Chem. News 61 (1890), 64.
2) Bull. de I'Academie roy. de medec. de Belgique. 1897.
The esters, which are nearly without exception of a pleasant odor, are often to be considered as the most important constituents of the oils. Thus, linalyl acetate is the carrier of the odor in bergamot oil; the same ester is found in lavender oil and also occurs along with other compounds in petitgrain oil.
The esters of borneol, found in the different pine-needle oils, play an important part in the formation of the pine aroma. Menthyl acetate is found in the peppermint oils and the geranyl ester of tiglic acid in the different geranium oils.
The quantitative estimation of the ester is always valuable for judging the oils, even when the esters are of little consequence to the odor. But it is much more important, and really the only rational test of quality in such cases where the esters are the carriers of the characteristic odor, as with bergamot oil and with lavender oil. The determination is made according to the method of quantitative saponification, as it has long been used in the analysis of the fats. Its application to the volatile oils was first urged by A. Kremel.1) However, it did not acquire practical importance until the nature of the saponifiable compounds was ascertained through scientific investigation.
Kremel distinguishes acid value (acid v.), ester value (ester v.), and saponification value (sap. v.). The acid value expresses how many mg. of KOH are necessary to neutralize the amount of free acid contained in 1 g. of oil. The ester value gives the amount of potassa in milligrams used in the saponification of the ester contained in 1 g. of oil. The saponification value is the sum of acid value and ester value. As the volatile oils with exception of e. g. geranium oil and vetiver oil usually contain only a small amount of free acid, this may in general be neglected. Only old, partly spoilt oils, tend to show somewhat higher acid values. It is urged that in all cases the acid and ester values be determined separately, for the reason that acids are occasionally added to increase the apparent ester content.
It should be mentioned that in all oils which contain aldehydes, the ester determination by saponification cannot be made, as a consumption of alkali takes place due to the decomposition of the aldehyde which increases with the length of the reaction, but which gives no information as to the amount of aldehyde decomposed.
1) Pharm. Post 21 (1888), 789, 821.
Errors in the determination likewise result from the presence of phenols. Hence these should first be removed with the aid of a dilute (3 to 5 percent) aqueous alkali, unless the ester determination is abandoned in such cases.
The saponification is conducted in a wide necked flask of potash glass of 100 cc. capacity (fig. 73). A glass tube about 1 m. in length and passing through a stopper serves as a reflux condenser. Into such a flask about 1,5 to 2 g. of oil are weighed accurately to 1 eg. and about twice its volume of acid-free alcohol is added. After the addition of a few drops of alcoholic phenol-phthalein solution, half-normal potassa is carefully added. The reaction is completed when the red color produced no longer disappears upon shaking. A later decoloration is due to the saponification of the ester by the alkali. After the neutralization of the free acid, which usually requires but 2 to 3 drops of halfnormal alkali, 10 cc. or more1) of the solution are added, and the mixture is heated on a water bath for an hour-) after the usual precaution to prevent bumping

Fig. 73.
1) In most cases 10 cc. suffice. Only in the case of oils with high ester content (Roman chamomile oil, wintergreen oil) 20 to 30 cc. should be taken. Care should always be taken to have a sufficient excess of alkali. For the same reason 20 cc. of alkali should be taken when oils of unknown ester content are to be saponified. If esters as such are to be saponified even larger amounts should be used depending upon the size of the molecule and the number of carboxyl groups present. Thus, e. g. the saponification of 2 g. of methyl formate requires 67 cc. of halfnormal potassium hydroxide solution.
2) The saponification of most esters can be accomplished in a much shorter time, that of the ester of bergamot oil being accomplished in 10 minutes. However, in order to be certain of the results and to work under the same conditions, the time of heating is extended whereby no harm is done. Comp. Report of Schimmel & Co. October 1895, 25. The method of Helbing (Helbing's Pharmacological Record Mo. 30, p. 4), according to which the saponification of bergamot oil is conducted in a closed vessel, hence under pressure, yields, as the investigations of Schimmel & Co. revealed, results that are 1 to 2 percent has been taken and the reflux condenser has been adjusted. After cooling, the contents of the flask are diluted with 50 cc. of water and the excess of alkali is titrated with half-normal sulphuric acid.
The computation is carried out with the aid of the following formula: in which a stands for the number of cc. of half-normal alcoholic potassa solution and s for the number of grams of oil of used.
in which a stands for the number of cc. of half-normal alcoholic potassa solution and s for the number of grams of oil of used.
If for 1,5 g. of bergamot oil employed 0,1 cc. of half-normal potassa has been used to neutralize the free acid, 6,0 cc. for saponification, then

From the ester value the percentage of ester or of alcohol respectively can be computed with the aid of the following formulas:
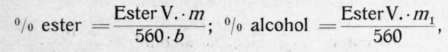 in which m stands for the molecular weight of the respective ester, m1 for that of the corresponding alcohol, and b for the basicity of the corresponding acid. • higher than those conducted in an open flask. The explanation lies in the fact that the linalool itself is affected by the caustic potash when heated with it under pressure whereas it suffers no change in the open flask. Later Helbing and Passmore [Chemist and Druggist 47 (1895), 585], substantiated the conclusion that the saponification in an open flask is to be preferred to that in an autoclave. Neither does the process of cold saponification appear to be applicable, which has the additional disadvantage of loss of time. According to Henriques (Zeitschr. f. angew. Chem. 1897, 399), linalool reveals a saponification value of 4,2, geraniol one of 2,8 when acted upon by alkali for 12 hours. In isolated cases only boiling for one hour does not suffice for the quantitative saponification of esters, thus e. g. with terpinyl acetate, bornyl and menthyl isovalerate, which must be boiled for from 2 to 3 hours with a sufficient excess of alkali.
in which m stands for the molecular weight of the respective ester, m1 for that of the corresponding alcohol, and b for the basicity of the corresponding acid. • higher than those conducted in an open flask. The explanation lies in the fact that the linalool itself is affected by the caustic potash when heated with it under pressure whereas it suffers no change in the open flask. Later Helbing and Passmore [Chemist and Druggist 47 (1895), 585], substantiated the conclusion that the saponification in an open flask is to be preferred to that in an autoclave. Neither does the process of cold saponification appear to be applicable, which has the additional disadvantage of loss of time. According to Henriques (Zeitschr. f. angew. Chem. 1897, 399), linalool reveals a saponification value of 4,2, geraniol one of 2,8 when acted upon by alkali for 12 hours. In isolated cases only boiling for one hour does not suffice for the quantitative saponification of esters, thus e. g. with terpinyl acetate, bornyl and menthyl isovalerate, which must be boiled for from 2 to 3 hours with a sufficient excess of alkali.
For the more common alcohols found in volatile oils, viz., C10H18O (geraniol, linalool, borneol, isopulegol), C10H20O (menthol, citronellol), C15H240 (santalol), C15H260 (cedrol), and their esters the formulas read as follows:

In the above formulas the factor b drops out since acetic acid is monobasic.
For the bergamot oil illustration given on the previous page, the ester of which is linalyl acetate (CH3COOC10H17) and the alcohol of which is linalool (C10H15O), the following values result:

Avoiding the ester value, the percentage of ester and of alcohol can be computed directly with the aid of the following formulas:
In these formulas the symbols have the same significance as in the previous formulas.
For the alcohols mentioned above, viz., C10H18O, C10H20O, C15H240 and C15H260 tables will be found at the end of this chapter (table I, p. 618), in which the percentage of acetic ester and of alcohol can be found for each ester value.1) The use of the table is quite apparent without further explanation. If in a given case 112 has been found as the ester value for bergamot oil, all that is necessary is to trace the corresponding figure in the column headed C10H18O and in the one headed "acetate", viz., 39,20. This figure reveals the percentage of linalyl acetate in bergamot oil, whereas the corresponding number 30,80 in the column headed "alcohol" reveals the percentage of linalool.
1) For geraniol the percentage of tiglate is also given. Table I, p. 618.
In order to save time in the computation of these alcohols, a second table (table II, p. 632) has been added in which the ester value (acid value, saponification value) as well as the corresponding percentage content of acetate or alcohol can be traced from the number of cc of half-normal potassium hydroxide solution used, provided exactly 1,50 g. of oil have been employed.
Continue to:


