II. Aromatic Series. Continued
Description
This section is from the book "A Manual of Materia Medica and Pharmacology", by David M. R. Culbreth. Also available from Amazon: Manual of Materia Medica and Pharmacology.
II. Aromatic Series. Continued
2. Incinerate 1 Gm
Incinerate Gm. ash .5 p. c. Impurities: Glucose, milk-sugar, ammonium compounds, salicylic acid, carbonizable substances. Dose, gr. 1-1 (.06-.26 Gm.).
Properties and Uses. - As a substitute for sugar, in diabetic or gouty patients; antiseptic, retards action of digestive ferments, hence should be used with caution where digestion is impaired. Sodium salt (soluble saccharin, gluside, crystallose) more soluble.
Acidum Phenylcinchoninicum. Phenylcinchoninic Acid, C6H5.C9-H5.N(COOH). - (Syn., Acid. Phenylcinch., Phenyl-quinoline-carboxylic Acid, Atophan; Ger. Phenylchinolincarbonsaure.) An organic acid, 2-phenylquinoline-4-carboxylic acid.
Manufacture: Heat for 3 hours on water-bath pyroracemic acid (1) and benzaldehyde (1), dissolved in alcohol, with aniline (1) previously dissolved in alcohol. It is in small, colorless needles, white or yellowish-white microcrystalline powder; odorless, or slight odor of benzoic acid; bitter taste; permanent; insoluble in water, slightly soluble in alcohol, hot water, ether, soluble in hot alcohol; melts at 210° C. (410° F.) with partial decomposition. Tests: 1. Saturated solution in hot diluted hydrochloric acid, + platinic chloride T. S. - reddish-brown precipitate (crystals). 2. Incinerate .5 Gm. - ash non-weighable.
3. Dissolve 1 Gm. in excess of ammonia water, evaporate to dryness on water-bath, add distilled water 20 Ml. (Cc); filtrate + silver nitrate T. S. - white, flocculent precipitate; + lead acetate T. S. - yellowish, flocculent precipitate; + copper sulphate T. S. - green, flocculent precipitate.
Properties and Uses. - Antirheumatic, antiseptic, analgesic, diuretic; gouty and rheumatic pains in joints or muscle sheaths, gouty and rheumatic neuralgia, neuritis, elimination of uric acid. Dose, gr. 5-20 (.3-1.3 Gm.), 4 times daily, preferably in alkaline water.
VI. Organic Bases.
Antipyrina. Antipyrine, C11H12N2O. - (Syn., Antipyr., Phenazone, Phenazonum, Methozine, Analgesine, Dimethylphenyl-isopyrazolone, Dehydrodimethylphenylpyrazine; Fr. Analgesine, Anodynine, Paro-dyne; Ger. Pyrazolonum phenyldimethylicum, Antipyrin.) This is phenyldimethylpyrazolon, obtained by the condensation of phenyl-hydrazine with aceto-acetic ether and methylation of the product.

Pyrole
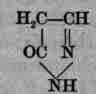
Pyrazolon

Antipyrine
Manufacture: Aniline chloride or sulphate is treated with sodium nitrite, the resulting diazobenzene chloride or sulphate is treated with reducing agents (alkali sulphite and stannous chloride), forming phenylhydrazine, C6H5HN.NH2, which is heated with aceto-acetic ether, CH3CO.CH2CO.OC2H5, producing phenylmethyl-isopyrazolone, C6H5N.CO.CH:C(CH3).NH; dissolve this in methyl alcohol, heat with methyl iodide, forming a compound which treated with sodium hydroxide solution splits off hydriodic acid and separates antipyrine, as a heavy oil, that may be crystallized from toluene or ether. It is a white, almost odorless, crystalline powder, tabular crystals, slightly bitter taste, soluble in water (1), alcohol (1.3), chloroform (1), ether (43); melts at 112° C. (234° F.); aqueous solution neutral, colorless. Tests: 1. Aqueous solution + tannic acid T. S. - abundant white precipitate. 2. Aqueous solution (1 in 100) 12 Ml. (Cc.) + sodium nitrite .1 Gm., + diluted sulphuric acid 1 Ml. (Cc.) - deep green (isonitroso-antipyrine). 3. Aqueous solution (1 in 1000) 2 Ml. (Cc.) + a drop of ferric chloride T. S. - deep red, changing to light yellow with 10 drops of sulphuric acid. 4. Incinerate 1 Gm. - ash .1 p. c. Impurities: Heavy metals, etc. Should be kept in well-closed containers. Dose, gr. 2-20 (.13-1.3 Gm.).
Properties and Uses. - Antiseptic, anodyne, antipyretic, cardiac depressant, haemostatic, diuretic, antineuralgic, analgesic, contracts the bloodvessels, migraine, locomotor ataxia, rheumatism, neuritis, urticaria, chorea, gout (arthritis), whooping-cough, tetanus, epilepsy, spasmodic laryngitis, sciatica, labor pains, diabetes, gravel, dysmenorrhoea, headache, angina pectoris, cancer, syphilitic pains, biliary and renal colic, skin diseases, eye affections, locally to ulcers and as a haemostatic. Antipyrine resembles acetanilid very closely, and owing to its larger dose should be handled with even more caution.
Poisoning: Similar to acetanilid.
Incompatibles: Iron chloride, iodide, and sulphate, copper sulphate, iodine, arsenic iodide, sodium bicarbonate, phenol (carbolic acid), hydrocyanic acid, nitric acid, nitrites, potassium permanganate, salicylates, calomel, corrosive sublimate, spirit nitrous ether. All tannin preparations give white precipitate, and hydrated chloral decomposes it.
Betaeucainae Hydrochloridum. Betaeucaine Hydrochloride. - (Syn., Betaeucain. Hydrochl., Eucaine Chloride, Eucaine; Fr. Chlorhydrate de Betaeucaine; Ger. Eucain B, Trimethylbenzoxy-piperidinhydrochlorid.)

Manufacture: This synthetic derivative of piperidine is made by treating diacetonamine with paraldehyde, reducing product with metallic sodium to vinyldiacetonalkamine, benzoylating the latter with benzoyl chloride, dissolving resulting free base, benzoylyinyl-diacetonalkamine, neutralizing with hydrochloric acid, crystallizing. It is a white crystalline powder, odorless, permanent, soluble in water (30), alcohol (35), chloroform (6); saturated aqueous solution neutral, + silver nitrate T. S. - white precipitate, insoluble in nitric acid; contains 99 p. c. of pure salt. Tests: 1. Dissolve .1 Gm. in distilled water (20), + a drop of ammonia water - white precipitate, dissolved by agitation; + 3 drops of ammonia water - white precipitate, dissolved by equal volume of distilled water. 2. Saturated aqueous solution + few drops of mercuric chloride T. S. - white curdy precipitate, or + few drops of a mixture of equal volumes of potassium chromate T. S. and diluted sulphuric acid - yellow curdy precipitate; incinerate .5 Gm. - ash non-weighable. Impurities: Cocaine, alpha-eucaine, readily carbonizable substances.
Properties and Uses. - Local anaesthetic; closely related to cocaine and tropacocaine. Has the advantage of cocaine in affecting much less the heart circulation and respiration, in not drying the corneal epithelium, nor causing mydriasis or disturbed accommodation; solutions may be sterilized by boiling water without decomposition, and are stable (dif. from cocaine); it has, however, the disadvantage of causing hyperaemia rather than contraction of mucous membranes when applied locally; largely employed in ophthalmology, 2-5 p. c. solution; for mucous surfaces, 2-5 p. c. Dose, hypodermically, ev-60 (1-4 Ml. (Cc.)) of 5 p. c. solution; may use ointment (1 p. c. + olive oil 2, wool fat 7).
Continue to:


