II. Aromatic Series. Part 8
Description
This section is from the book "A Manual of Materia Medica and Pharmacology", by David M. R. Culbreth. Also available from Amazon: Manual of Materia Medica and Pharmacology.
II. Aromatic Series. Part 8
Uses. - Phthisis, typhoid fever, hyperpyrexia, sthenic fevers, gas-tralgia, locomotor ataxia, rheumatism, sciatica, lumbago, neuralgia, gangrene, cancer, headache, insomnia, epilepsy, whooping-cough. There are many proprietary preparations containing 50-90 p. c. of acetanilid with sodium bicarbonate, ammonium or sodium bromide, salicylic acid, etc.
Poisoning: Have sweating, depression, slow breathing, irregular pulse, collapse, vomiting, cyanosis, prostration, death. Empty stomach, place in recumbent position, loosen clothing, plenty fresh air (for cyanosis), give diffusible stimulants (brandy, whisky, alcohol, ammonia, ether injections), coffee, atropine, strychnine (hypodermic), heat to feet and body, oxygen inhalations, rub skin, castor oil.
Incompatibles: Caustic soda and potash, chloroform.
Acetphenetidinum. Acetphenetidin. - (Syn., Acetphen., Phen-acetin, Phenacetinum, Para-acetphenetidin, Para-ethoxyacetanilide; Fr. Amido-acetique de-1'amido-phenetol; Ger. Phenacetin.)

Manufacture: This monoacetyl derivative of para-amidophenetol is obtained by acting on phenol with diluted nitric acid, producing ortho- and para-nitrophenol - C6H5OH + HNO3 = C6H4(NO2)OH + H2O; as para-nitrophenol is non-volatile, the two are separated by distillation with steam, the residuary para- being decolorized, crystallized and treated with sodium hydroxide, forming sodium nitrophenol, C6H4(NO2)ONa, which, by heating with ethyl iodide, is converted into para-nitrophenetol - C6H4(NO2)ONa + C2H5I = C6H4(NO2)OC2H5 + Nal - which, by the action of nascent hydrogen, is converted into paraphenetidin - C6H4(NO2)OC2H5 + H6 = C6H4-(NH2)OC2H5 + 2H2O, which, by prolonged boiling with glacial acetic acid, becomes para-acetphenetidin, similar to the conversion of aniline into acetanilid. It is in white, glistening, crystalline scales or fine powder; odorless; slightly bitter taste, faintly numbing the tongue; permanent, melts at 133° C. (272° F.); soluble in water (1310), boiling water (82), alcohol (15), boiling alcohol (2.8), chloroform (14), ether (90); aqueous solution neutral. Tests: 1. Boil .1 Gm. with hydrochloric acid (1), + distilled water (10), cool, filter, + a drop of potassium dichromate T. S. - ruby-red. 2. Incinerate 2 Gm. - ash .05 p. c. Impurities: Acetanilid, para-phenetidin, readily carbonizable substances. Dose, gr. 3-10 (.2-.6 Gm.).
Properties and Uses. - Antipyretic, analgesic, cardiac depressant, diuretic, safer than antipyrine or acetanilid; neuralgia, sciatica, locomotor ataxia, migraine, headache, hysteria, asthma, whooping-cough, epilepsy, rheumatism, chronic myelitis, arthritis, cystitis, metritis, acute neuritis, pleurisy, dysmenorrhoea. For any paroxysm of headache, etc., give gr. 3 (.2 Gm.) every hour until 3 or 4 doses are taken; it is decomposed in the system into phenetidin and amidophenol, and is eliminated chiefly by the kidneys.
Poisoning: Excessive quantity may produce vomiting, sweating, feeble and rapid pulse, collapse; treat as for acetanilid, with alcoholic stimulants, strychnine hypodermically, warmth externally, etc.
Methyl Phenacetin, C6H4C2H5CH3C2H3O, and Ethyl Phenacetin, C6H4OC2H5C2H5C2H3O, are also hypnotics, the first being the stronger.
Benzosulphinidum. Benzosulphinide. - (Syn., Benzo-sulphinid., Saccharin, Glusidum, Gluside, Neosaccharin, Glucusimide, Anhydro-orthosulphamidebenzoic Acid; Fr. Acide Anhydro-Orthosulfamide-Benzoique; Ger. Benzoesauresulfimid, Zuckerin, Toluolsiiss, Saccharol, Sycose.)
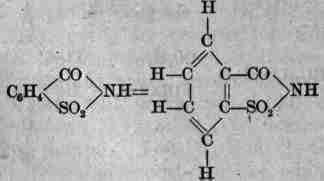
Manufacture: This anhydride of ortho-sulphamide-benzoic acid (benzoyl sulphonicimide) is prepared by a process involving six different steps: Take C6H5CH3 (toluene) + H2SO4 + 100° C. (212° F.) = C6H4(CH3)SO3H (ortho- and para-toluenesulphonic acids), which are converted into calcium and then sodium salts by sodium carbonate; act upon these with • phosphorus pentachloride = ortho- and para-toluenesulphochlorides, C6H4(CH3)SO2C1, cool, when para- crystallizes out; to ortho- add ammonia gas = ortho-toluenesulphamide, C6H4-(CH3)SO2NH2, which is oxidized with potassium permanganate = potassium ortho-sulphamidebenzoate, and this freed from precipitated manganese dioxide and decomposed by an acid splits up into its aldehyde and water, instead of separating as free ortho-sulphamidebenzoic acid. It is in white crystals, white crystalline powder, odorless or faintly aromatic, taste intensely sweet in dilute solution, soluble in water (290), boiling water (25), alcohol (31), ammonia water, alkali hydroxide solutions, sodium bicarbonate solution with evolution of carbon dioxide, slightly soluble in chloroform, ether; saturated aqueous solution acid; melts at 220° C. (428° F.). Tests: 1. Fuse gently 1 Gm. with sodium hydroxide (5) - ammonia vapors evolved; after these cease dissolve residue in distilled water (10), neutralize with diluted hydrochloric acid, filtrate + a drop of ferric chloride T. S. - violet.
Continue to:


