Atomic Theory
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Atomic Theory
Atomic Theory, the doctrine that matter consists of ultimate particles or atoms incapable of division. This idea was first maintained speculatively in opposition to the notion that matter is capable of being divided to infinity. Modern science has adopted this idea, not merely as a speculation which cannot be verified, but as a proposition which interprets and harmonizes a wide range of experimental facts. Inasmuch as it offers an explanation of the facts and principles of chemistry, these require to be noticed before we can understand the use and necessity of the theory. Modern chemistry took its rise with the abandonment of the old notion of phlogiston, and the elucidation of the principles of combustion by Lavoisier. He introduced the balance as a fundamental instrument of chemical inquiry, and thus placed the science upon a firm quantitative basis. As weighing became general and accurate, it was soon discovered that chemical combination is definite, and chemical composition constant. A certain weight of alkali, for example, combines with a given weight of acid to produce a salt, which therefore has a fixed numerical constitution. A great number of experiments showed that chemical union always takes place in this manner, and thus was established the fundamental law of definite proportions.
It was next discovered that combination may take place between the same substances in different proportions, and that when this is the case these proportions have simple numerical relations to each other. Thus, if two elements A and B are capable of uniting in several proportions, they may be represented as A + B, A + 2B, A+ 3B, A+ 4B, etc. The relations are not always so simple as this, but the principle is general, and is known as the law of multiple proportions. Again, it was found that if two elements which combine with each other combine also with a third, the proportions in the first combination are preserved also in the second. If a body A unites with certain other bodies B, C, D, then the quantities B, C, D, which combine with A, or certain simple multiples of them, represent for the most part the proportions in which they can unite among themselves. This is known as the law of equivalent proportions or chemical equivalence. It having thus been found that chemical actions follow strict numerical methods, and that each body has its fixed measure, it became important to determine exactly what these measures are.
This resulted in the scale of combining numbers or equivalents, or, as they are now more commonly termed, atomic weights, which constitute the foundation of the science and are given in all text books. - But if all kinds of matter in their chemical transformations are ruled by these numerical principles, we should expect that other material properties would be affected by them, and such is the fact. The combining weights of those elements which are known to exist in the state of gas or vapor are, with one or two exceptions, proportional to their specific gravities in the same state. Thus, the specific gravity of hydrogen being 1, that of oxygen is 16, sulphur vapor 32, chlorine 35.5, iodine vapor 127; but the figures represent also the combining numbers of these elements. Mr. Watts thus expresses the law of combination by volume: " If the smallest volume of a gaseous element that can enter into combination be called the combining volume of that element, the law of combination may be expressed as follows: The combining volumes of all elementary gases are equal, excepting those of phosphorus and arsenic, which are only half those of the other elements in the gaseous state; and those of mercury and cadmium, which are double those of the other elements." Gay-Lussac showed that combinations by volume take place in definite and multiple proportions, and that the volume of a compound gas always bears a simple ratio to the volumes of its elements, thus:
1 vol. hydrogen and 1 chlorine form 2 vols, hydrochloric acid.
2 vols. " " 1 oxygen " 2 " watery vapor.
3 " " " 1 nitrogen " 2 " ammonia.
Again, it is found that in many cases two or more compounds which are supposed to contain equal numbers of equivalents of their respective elements crystallize in the same or in very similar forms, and such compounds are said to be isomorphous. Accordingly, these isomorphous relations are often appealed to for the purpose of fixing the constitution of compounds, and thence deducing the atomic weights of their elements, in cases which would otherwise be doubtful. It has also been established that substances having different properties may have the same relative proportion of constituents, and such are said to be isomeric. Moreover, something analogous to this is seen among the elements themselves: they are capable of assuming different states, which capability is called allotropism. In both cases we are compelled to assume that their constituent parts are subject to differences of arrangement. Combining quantities are also intimately related to heat. This relation is thus stated by Mr. Watts: "The atomic weights of the elements, determined according to their modes of combination, are for the most part inversely proportional to their specific heats; so that the product of the specific heat into the atomic weight is a constant quantity.
The same quantity of heat is required to produce a given change of temperature in 7 grains of lithium, 5(5 of iron, 207 of lead, 108 of silver, 196.7 of gold." Finally, the law of combining proportions is implicated with the electrical relations of matter. Prof. Faraday proved that an equivalent of an element consumed in a battery gives rise to a definite quantity of electricity, which will produce exactly an equivalent of chemical decomposition. For example, the consumption of 32 grains of zinc in a battery excites a current which will set free from combination 1 grain of hydrogen, 108 of silver, and 39 of potassium; these being the combining numbers of the respective elements. - The facts above stated are independent of all hypothesis, and are the results of pure experiment. They demonstrate that in its ultimate and minutest form matter is in some way numerically constituted. How it is constituted was a question which the human mind could not escape. It was necessary to frame some clear conception of its ultimate constitution that would connect and interpret the known facts.
This was done by Dr. John Dalton of Manchester, England, in constructing the atomic theory, He was aware of the law of definite proportions, and he discovered the law of multiple proportions by investigation of the compounds of carbon and hydrogen, of oxygen and carbon, and of nitrogen and oxygen. To account for these laws, he assumed, first, that all matter consists of indivisible, unchangeable atoms of extreme minuteness; second, that all the atoms of the same element have the same weight, but that in different elements they have different weights; third, that these relative weights correspond with the combining numbers, which may therefore be called atomic weights; fourth, that these different atoms have mutual attractions and combine to form chemical compounds, not by interpenetration of their substance, but by atomic juxtaposition. If this idea be admitted, the principles of chemical constancy and definite proportions follow as inevitable consequences. The definite proportions in which bodies combine represent the constant ratio between the weights of the combining atoms. The principle of multiple proportions is equally explained, for the successive additions must be made by whole atoms, and therefore by whole numbers.
One atom of carbon unites with one atom of oxygen to form carbon monoxide, and with two atoms of oxygen to form carbon dioxide. That the atomic weights of compounds must equal the sum of the atomic weights of their elements follows with equal certainty. Moreover, in the rearrangement of atoms in a body, without addition or subtraction of elements, we have a ready explanation of isomeric and allo-tropic changes. The relations of chemical changes to heat, now expressed by the phrase "atomic heat," and their relation to volume, indicated by the phrase "atomic volume," become in like manner capable of explanation on the assumptions of the atomic theory. It is a merit and a test of this theory that its resources have kept pace with the rapid extension of the science, but it has required to be itself developed for this purpose. In the hands of Dalton it was applied to a few simple fundamental facts; it now embraces facts of many orders and of greater complication. At present the conception of the molecule or the group of combined atoms plays a much more important part than it did at first. Even the atoms of the elements (as will be presently explained) are now conceived not to exist separately, or as units, but as combined with each other in a molecular condition.
An atom is defined as the smallest particle of simple matter that can enter into the composition of a molecule. A molecule is defined as a group of atoms held together by chemical force, and is the smallest particle of any substance that can exist in a free or uncombined state in nature. Molecules are of two kinds: elemental molecules, in which the atoms are alike, and compound molecules, in which the atoms are unlike. Molecular structure, the outgrowth of the conception of atoms, is now the fundamental idea by which chemistry and physics are connected. - The doctrine of Dalton at first seemed to afford an easy explanation of chemical equivalents, by which one body may replace another, or be substituted for it by simple exchange of atoms. But recent discoveries have shown that it fails here and requires extension. It was formerly supposed that when one element replaces another in a combination, the substitution always takes place atom for atom, and hence the terms atom and equivalent were regarded as synonymous.
But it is now known that this is only true for certain elements, which are accordingly classed as monogenic elements. There are others which always take the place of two or more atoms of a monogenic element, and these are termed polygenic elements. This brings us to the new conception of atomicity, which has now become the fundamental idea of the science. To understand it properly, it will be necessary to glance at the steps of chemical theory by which it has been reached. The name of Lavoisier is intimately associated with the first general theory of chemical combination. This was the binary or dual system of chemistry. An acid was held to result from the union of a simple body (generally non-metallic) with oxygen; an oxide resulted from the combination of oxygen with a metal; a salt was produced by the union of an acid with an oxide, and this pairing of doubles represents its constitution. In all combinations affinity is assumed to be exerted upon two elements, simple or compound, which attract one another and unite by virtue of opposite properties, all chemical compounds being therefore binary. This is dualism, and the chemical nomenclature was constructed upon the idea.
The view proposed by Lavoisier was ably enforced by Berzelius. Electro-chemistry, by which bodies were decomposed into pairs that appeared at opposite poles of the battery, lent powerful aid to the binary theory; and Berzelius carried it out by arranging the elements on a scale of antithesis as electro-positive and electro-negative. In 1816 he also devised a new notation, now in general use, by which letters symbolize the elements, and composition can be compendiously represented to the eye by means of formulas. Prof. Wurtz, in his "History of Chemical Theory," says: "By the arrangement of these formulas in which the acid appeared on one side with the train of oxygen atoms belonging to it, and the metallic base on the other with the oxygen united to the metal, Berzelius gave to the dualistic system a degree of precision unknown before his time." But a true scientific theory must embrace all orders of facts to which it is applicable. Dualism was well fortified in mineral chemistry, but it was not easy to bring the complexities of organic chemistry into harmony with it. Berzelius, however, made this his great task. There were organic acids, organic bases, and organic salts; and these were represented on the binary plan.
Organic radicals were also discovered - compounds which played the part of simple elements; and these were subordinated to the binary system.. By this theory of compound radicals dualism was extended to organic chemistry, and chemical theory was apparently unified. Yet the victory was far from complete. The deeper study of organic compounds led eminent chemists to question the validity of the dual hypothesis as applied to them. A school arose led by Dumas, Laurent, and Ger-hardt, which took a new view of the constitution of organic bodies. Its first idea was the doctrine of substitutions, and in its application a breach was made at the outset in the electrochemical theory. It was found that chlorine, a powerful electro-negative element, could replace hydrogen, a strong electro-positive element, in an organic compound, playing the same part and not altering the character of the compound. The new view, rejecting dualism, regarded organic bodies as units, or as unitary structures; and their changes by substitution were likened to the alteration of an edifice by successively removing its individual bricks and stones and replacing them by others.
Laurent compared organic compounds to crystals, whose angles and edges may be replaced by new atoms or groups of atoms, while the typical form is preserved. Thus to the dualistic point of view was opposed the unitary system; to the idea of combination resulting from addition of elements was opposed that of compounds formed by substitution of elements. An acid is changed to a salt by substituting a metal for its hydrogen, without destroying its molecular structure. A salt is no longer to be regarded as a binary compound, containing an acid on the one side and an oxide on the other; it is a whole, a single group of atoms, among which are one or more atoms of metal capable of being exchanged for other metallic atoms or for hydrogen. This view led to the theory of chemical types, in which certain substances are taken as patterns of molecular structure with which analogous bodies are classified. Thus we have the water type, the hydrogen type, and the ammonia type, under which bodies are grouped with no reference to their former relationships.
The binary theory here disappears, and substances are brought together not so much on the principle of composition or atomic arrangement, as by analogies of reaction and decomposition. - But the doctrine of types was transitional, and soon developed into the completer theory of atomicity, by which is meant combining capacity. For example, there are some acids which require for saturation only one equivalent of a certain base; there are others which require two equivalents of the same base to saturate them; and others still which demand three. Now these acids are clearly not equivalents of each other, their capacities of combination varying as 1, 2, 3; and they are therefore said to have different atomicities. This conception of the varying combining powers of bodies, as a controlling chemical principle, was worked out in the field of organic chemistry; but it is now extended to the inorganic elements, and offers a new system of classification and a new chemical method. - In the new chemistry the elements are arranged into six groups, although some add a seventh. These are named monads, dyads, triads, tetrads, pentads, and hex-ads - terms expressive of their several combining capacities.
Monads, of which hydrogen, chlorine, and potassium are examples, are monogenic, that is, they can combine only with single atoms. All the rest are polygenic, that is, they can combine with 2, 8, 4, 5, or 6 monogenic elements or their equivalents. Molecules are also designated as monatomic, diatomic, triatomic, tetratomic, pentatomic, and hexatomic. For equivalence, which represented the old idea, the term valence is coming into use; and a series of words is derived from it describing the groups as univalent, bivalent, trivalent, quadrivalent, quinquivalent, and sexi-valent, while the atomicities above univalence are termed multivalent. The varying equivalence, valence, or combining power of atoms is represented in several ways by which the idea is made clear. The graphic symbol of an atom is a circle with lines radiating from it, called bonds, which indicate the valence or atomicity. They are represented as follows, the first line giving their names, the second their symbols, and the third examples:
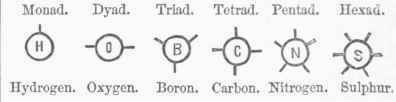
Water, OH2, would be thus represented by graphic formula:
Hydrogen.
has as it were but a single pole of attraction, represented by a single bond, while oxygen has two poles and two bonds. The attractions of the two atoms of monatomic hydrogen are satisfied by the two attractions of diatomic oxygen. So carbon-dioxide, CO2, may be represented thus:
Here the four attractions of tetratomic carbon are saturated by those of the two atoms of diatomic oxygen. Marsh gas, CH4, is thus represented: The circle may be omitted, and the bonds connected directly frith the letters, thus, it being immaterial how the bonds are arranged. The composition of water will then be represented thus, H - O - H, and carbon-dioxide 0 = C=0. The atomicity is often represented as follows by dashes: IF, 0", B'", C"", N'"", S"""; or again thus by Roman numerals: H1, O11, B111, CIV, Nv, SVI. In chemical changes and the formation of new compounds all attractions require to be satisfied - every bond engaged. This fact fixes a limit to combination, for certain groupings become impossible. One atom of a monad cannot unite with one atom of a dyad, because one attraction cannot neutralize two. It takes two atoms of a monad to form a compound with an atom of a dyad; four atoms of a monad or two atoms of a dyad are required to saturate a tetrad; but in each case all the polarities have to be provided for.
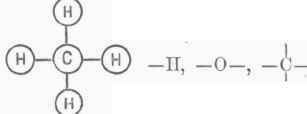
There are seeming exceptions to this law. Two atoms of a monad element, as potassium, may unite with one, two, three, four, or five atoms of a polyad element, as sulphur. By an examination of the graphic formulas of those compounds, K - S - K, K - S - S - K, K - S - S - S - K, etc, it is seen that any number of atoms of a polyad element may • unite with two atoms of a monad, provided they be interposed between the latter. When thus placed, they are said to perform a linking function in the compound. The atomicity of an element is its highest equivalence, and the compound form is then said to be normal or saturated. Yet the equivalence of atoms is not always the same; an atom may form several compounds of the same substance. Elements of even equivalence, in which the atomic poles are in pairs, are called artiads; those of uneven equivalence, in which the poles are odd, are termed perissads. Prof. Barker states that the equivalence "always increases or diminishes by two; so that an atom of the same element may in different compounds have an equivalence of 1, 3, 5, or 7, or of 2, 4, or 6. A perissad atom can never become an artiad atom by such a change, nor can an artiad become a perissad." This variation of atomic equivalence is accounted for on the hypothesis that the bonds of an atom are capable of saturating each other in pairs.
A pentad may thus become a triad and a monad successively, and a hexad may be converted into a tetrad or into a dyad, as follows:

It follows from this view that only the atoms of those free elements can be considered as existing separately in which the number of bonds is even. The others can only exist in combination with each other, forming polyatomic molecules. Free hydrogen cannot be - II, because its bond is unsatisfied; it must therefore be H - H, that is, united with itself, forming what we might call hydride of hydrogen. Chlorine is not CI - , but CI - CI, and free oxygen is not - 0 - , but 0=0. Compounds are formed by replacement, and chemical science thus becomes rooted in atomic capacity. - While therefore in the last quarter of a century chemical philosophy has undergone a total revolution, the atomic theory has not only been maintained and strengthened, but it is doubtful if the advance could have been made without its assistance.
Continue to:


