Phosphorus
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Phosphorus
Phosphorus (Gr. ![]() , light, and
, light, and ![]() , to carry), an elementary body, discovered by Brandt of Hamburg in 1669, in the solid residue left on evaporating urine, while attempting to obtain a liquid capable of transmuting silver into gold. Kunckelj a German chemist, learned the source of the new substance and communicated the information to Kraft of Dresden, who went to Hamburg and paid 200 dollars for the details of the process. In the mean time Kunckel succeeded in preparing phosphorus. In 1680 it was noticed in the " Philosophical Transactions " of the royal society of London, and it was soon prepared in considerable quantities by Godfrey Hank-witz under the direction of Robert Boyle. In 1737 the French government purchased and published a method of preparing phosphorus from urine; but the unpleasant and expensive process was abandoned after Gahn in 1769 discovered it as a constituent of bones, and Scheele six years later devised a process for its extraction, by which burnt bones were digested with dilute nitric acid, the lime precipitated by sulphuric acid, and the filtrate evaporated to a sirup, which Was then mixed with charcoal powder and subjected to distillation, phosphorus being set free and coming over as a vapor, which was condensed in water.
, to carry), an elementary body, discovered by Brandt of Hamburg in 1669, in the solid residue left on evaporating urine, while attempting to obtain a liquid capable of transmuting silver into gold. Kunckelj a German chemist, learned the source of the new substance and communicated the information to Kraft of Dresden, who went to Hamburg and paid 200 dollars for the details of the process. In the mean time Kunckel succeeded in preparing phosphorus. In 1680 it was noticed in the " Philosophical Transactions " of the royal society of London, and it was soon prepared in considerable quantities by Godfrey Hank-witz under the direction of Robert Boyle. In 1737 the French government purchased and published a method of preparing phosphorus from urine; but the unpleasant and expensive process was abandoned after Gahn in 1769 discovered it as a constituent of bones, and Scheele six years later devised a process for its extraction, by which burnt bones were digested with dilute nitric acid, the lime precipitated by sulphuric acid, and the filtrate evaporated to a sirup, which Was then mixed with charcoal powder and subjected to distillation, phosphorus being set free and coming over as a vapor, which was condensed in water.
It was afterward found to be a constituent of certain primitive rocks, especially of the mineral apatite. (See Lime, vol. x., p. 478.) The decomposition of these rocks furnishes phosphorus in the form of phosphate of calcium to the soil, from which it is appropriated by plants, these in turn supplying the animals which feed upon them, for the building of whose structures, particularly the bones and the nervous system, it is indispensable. Phosphorus is now prepared from bones and also from the native calcic phosphate. (For the chemical composition of bones, see Bone, and Bone Dust.) In the preparation of phosphorus the bones were formerly calcined in an open fire, but now the gelatine is first extracted by water with the aid of heat and pressure, or they are first made into bone black, which after being employed for refining sugar is burned. Three parts of powdered bone ash are mixed with two of concentrated sulphuric acid, or three parts of the crude acid of specific gravities 1.55 and 18 or 20 parts of water. After standing two or three days, the mixture is placed in a strong linen bag, and the liquid containing the phosphoric acid is separated from the sulphate of lime (calcic sulphate) by pressure. Water is added to the residue, and the washings are added to the filtrate.
The sulphuric acid should be just sufficient to remove two thirds of the calcium, leaving the remainder as a soluble acid salt combined with all the phosphoric acid, which is often called superphosphate of lime. The reaction is shown in the following equation:
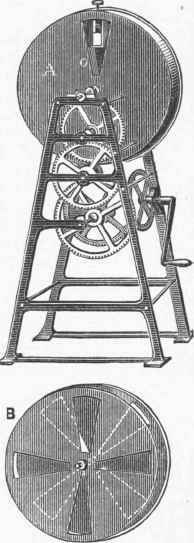
Phosphoroscope.
Ca32P04 + 2H2S04 = H4Ca2P04 + 2CaS04.
Tricalcic phos- Sulph. acid. Acid calcic phos- Calcic sulphate, phate. phate.
The acid solution, after being evaporated to a sirupy form, is mixed with one third or one half its weight of charcoal and raised to a nearly red heat in an iron pot, and then transferred while hot to an earthen retort, a, fig. 1. It is then gradually heated to full redness, by which means the carbon combines with the calcium base and with oxygen to produce water, tricalcic phosphate, and phosphorus.
This reaction is divided into two stages. The superphosphate is first decomposed into water and calcic metaphosphate (H4Ca2P04=Ca 2P03 + 2H20), and the metaphosphate, by the action of carbon, is reconverted into tricalcic phosphate (bone phosphate), carbonic oxide, and phosphorus, the two latter passing over in a gaseous form (3Ca2PO3 + 10C=4P + Ca32PO4 + 10CO). The phosphorus may be purified by fusing it under water containing some bleaching powder, and squeezing through wash leather. It is moulded in the form of sticks by raising it in glass tubes by atmospheric pressure. - Properties. Phosphorus is a soft, translucent, colorless solid, of a waxy consistency, becoming brittle at low temperatures. Its symbol is P, its atomic weight 31, and its specific gravity when solid 1-83, when liquid below its melting point 1.76 (Gladstone and Dale); its observed vapor density is 4.42; its melting point 111.5° F. If melted under an alkaline liquid and cooled slowly, it will remain for some time fluid at ordinary temperatures, but when touched with a rod will suddenly solidify. Its boiling point is 550°, an atom of its vapor occupying once and a half as much space as an atom of hydrogen.
According to Deville, this relation of volumes is not affected by a temperature of 1904°. When melted phosphorus is slowly cooled, well formed dodecahedrons may be obtained. It is insoluble in water, for which reason it is usually preserved in that liquid; is soluble in oil, in petroleum naphtha, and in bisulphide of carbon. It is exceedingly inflammable, taking fire by the heat of the hand, and a blow will often kindle it. In the open air it burns with a bright flame, forming with oxygen phosphoric acid. At ordinary temperatures a stick of phosphorus emits a white smoke, which is luminous in the dark, in consequence of slow combustion, and upon this action depends one of the methods of analyzing the air. (See Ni-teogen.) This slow combustion (oxidation) may be prevented by a small quantity of ole-fiant gas, vapor of ether, or some essential oil. It is remarkable that in pure oxygen phosphorus is not luminous until the temperature is as high as 59°, or unless the gas is rarefied, or diluted with nitrogen, hydrogen, or carbonic acid. Phosphorus assumes several different forms. The translucent variety, common or vitreous phosphorus, has been described.
When this is exposed to light under water, it becomes white and opaque, somewhat less fusible, and of less specific gravity (1.515). This variety becomes reconverted into the common form at a temperature of 122° F. A third form, black and opaque, is obtained by suddenly cooling melted phosphorus, which by fusion and slow cooling returns again to the common form. A viscous form, analogous to viscous sulphur, is produced by suddenly cooling phosphorus heated to near its boiling point. A fifth form occurs in red scales, and is known as red or amorphous phosphorus. It has been carefully examined by Schrotter, and may be obtained by exposing phosphorus in a vacuum to the rays of the sun, or by subjecting it for 50 hours to a temperature of 446° to 464° F. in an atmosphere of some gas with which it does not combine. This red, amorphous, opaque variety is insoluble in bisulphide of carbon or benzole, and when exposed to the air emits no odor. Its density is greater than that of common phosphorus, being from 2.089 to 2.106. It remains unchanged in the open air till heated to 500°, when it melts and bursts into a flame, emitting dense clouds of phosphoric anhydride. When slightly rubbed with chlorate of potash, peroxide of manganese, or peroxide of lead, it detonates and inflames.
On account of its property of remaining unchanged at ordinary temperatures, but of combining with the oxygen of oxygen compounds by means of friction, it is used advantageously in the manufacture of friction matches. For this purpose it may be mixed with the other materials, or with size and pounded glass and spread on paper, the match, tipped with chlorate of potash and sulphur, being rubbed upon it. Common or vitreous phosphorus taken into the stomach acts as a powerful irritant poison, but the amorphous variety may be swallowed with impunity. Those who work in common phosphorus are liable to be attacked with necrosis of the jaw bones. (See Match, and Necrosis.) - Corn-founds. With oxygen phosphorus forms two definite oxides, in which the relative quantities of oxygen are as 3 to 5. They are phosphorous anhydride, P203, and phosphoric anhydride, P2O5. It also forms three oxidized acids: hypo-phosphorous acid (monobasic), HPH202; phosphorous acid (dibasic), H2PH03; and phosphoric acid (tribasic), H3P04. The most important of the oxides, phosphoric anhydride, is that which forms phosphoric acid by uniting with water. It occurs in the native tricalcic phosphate (apatite) and in bone phosphate.
It may be pre-. pared by burning dry phosphorus in a glass vessel to which a supply of dry air is admitted. A, fig. 2, is a glass globe with a vessel, B, containing ignited phosphorus, suspended by a platinum wire from the tube H, through which phosphorus may be added, and also ignited by means of a hot wire. 0 is a drying tube, containing chloride of calcium, or pumice moistened with oil of vitriol. A bent tube, D, leads into the bottle E, from which proceeds another tube, F, connected with an aspirator for drawing air through the apparatus. Phosphoric anhydride has a powerful attraction for water. Exposed to the air for a few moments, it absorbs moisture and becomes liquid, and when thrown into water combines with it, with explosive violence, being converted into phosphoric acid, and the water is so firmly bound that it cannot be separated without ultimate decomposition (P205 + 3H20=2H3P04). It is powerfully dehydrating, abstracting water from acids, alcohols, and other bodies, and reducing oil of vitriol to an anhydride, and is one of the most powerful desiccating agents known. The pure acid is usually produced in a hydrated state by boiling one part Of phosphorus in about 13 parts of nitric acid of specific gravity 1'2, which furnishes the oxygen, while nitric oxide is evolved.
The nitric acid being mostly distilled off, the residue containing the phosphoric acid is transferred to a platinum vessel and cautiously heated to redness. This expels a portion of the water of hydration, leaving the acid in the form of a monohydrate, or the glacial phosphoric acid (metaphosphoric acid) of the pharmacopoeia. There, are three distinct forms of phosphoric acid, a fact first shown by Graham, viz.: metaphosphoric acid, HP03; orthophosphoric acid, H3P04; and pyrophosphoric acid, H4P207. These different forms retain their characteristics when dissolved in water, and form salts with one, with three, and with four equivalents of metals, which are respectively called meta-phosphates, orthophosphates, and pyrophosphates. The study of these compounds has greatly assisted in forming the modern theory of saline compounds in general. Of these salts the orthophosphates are the most common, and the most familiar example is the tricalcic phosphate, found in bones and in the mineral apatite, Ca32P04. Among other common or orthophosphates are trisodic phosphate, Na3P04, 12H20; sodic dihydric phosphate, NaH2P04, H20; and triargentic phosphate, Ag3P04. Among the metaphosphates are sodic meta-phosphate, NaP03, and argentic metaphos-phate, AgP03; and among the pyrophosphates, sodic pyrophosphate, Na4P2O7,10H2O, and argentic pyrophosphate, Ag4P207. Phosphorous anhydride, P203, may be obtained by burning phosphorus in a limited supply of dry air, by which a white volatile, deliquescent, inflammable powder is produced.
The acid derived from this, phosphorous acid, H2PH03, is formed by the union of one molecule of P203 with three of water (P203 + 3HaO=2H2PH03), and may be obtained by passing a stream of chlorine very slowly through a deep layer of phosphorus melted under water, so that each bubble of gas shall be completely absorbed by the phosphorus. Chloride of phosphorus, PC13 is formed, arid is immediately decomposed by water into hydrochloric and phosphorous acids (P013 + 3H2O=H2PHO3 + 3HC1). By concentrating the acid liquid by heat not exceeding 390°, the hydrochloric acid is expelled and the phosphorous acid obtained in deliquescent rectangular prisms. Phosphorous acid is dibasic, and forms two classes of salts called phosphites. The normal salts have the general formula M2PH03, and the acid salts MHPHO3, where M represents the metallic element. - Hypophos-phorous acid, HPH202, is not derived from any known phosphorus anhydride. When phosphorus is boiled in a caustic alkaline solution, or with a hydrate of one of the alkaline earths, a hypophosphite is formed with evolution of phosphuretted hydrogen, as mentioned in describing phosphuretted hydrogen.
By evaporation the hypophosphite may be obtained, and by adding to it sulphuric acid the hypophos-phorous acid may be obtained in solution; while if lime or baryta be used, an insoluble sulphate is separated. The salts formed by the union of this acid with bases are called hy-pophosphites, and they have been shown by the researches of Dulong, Rose, and Wurtz to be monobasic, and therefore constitute but a single monobasic class, of which the hypophosphite of soda, NaPH202, may be regarded as the type. The hypophosphites are all soluble in water and generally crystallize easily, but when the evaporation takes place at a high temperature they are converted into phosphites by absorption of oxygen. - With hydrogen phosphorus forms three compounds (phosphides or phospburets), PH3, PH2, and P2H. The first is a gas, the second a liquid, and the third a solid at ordinary temperatures. The gaseous phosphide, common phosphuretted hydrogen, may be obtained pure by heating phosphorous acid in a retort, phosphoric acid and phosphuretted hydrogen being produced (4H3P03 = PH3 + 3H3P04). The gas has a density of 1.24, every two volumes containing three volumes of hydrogen and a half volume of phosphorus vapor.
It has a disagreeable garlic odor, is slightly soluble in water, and burns with a brilliant white flame, forming water and phosphoric acid. Phosphuretted hydrogen may also be produced by boiling phosphorus in milk of lime or a solution of caustic alkali. The retort or flask should be very nearly filled, and a glass tube leading from it should dip under the surface of water to avoid explosions. The reaction is represented in the following equation:
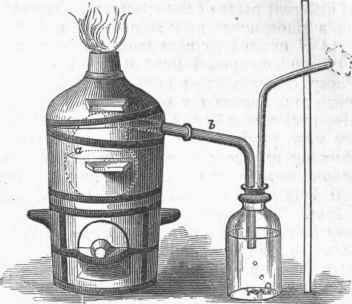
Fig. 1. - Manufacture of Phosphorus.
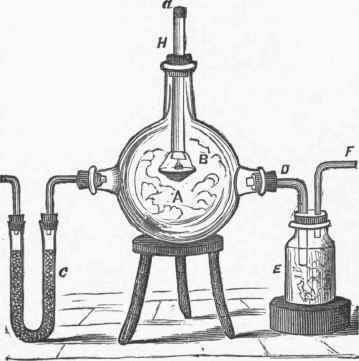
Fig. 2. - Formation of Phosphoric Anhydride.
8P + 30aH202 + 6H20 = 2PH3 + 3CaH4P204.
Calcic Water. Phosph. Calcic hypohydrate, hydr. phosphite.
Calcic hypophosphite and phosphuretted hydrogen are generated. Prepared by this process, the gas has the remarkable property of inflaming spontaneously in the presence of oxygen or atmospheric air. As the bubbles come to the surface they inflame and produce beautiful rings of phosphoric acid, as shown in fig. 3, which also shows the arrangement for preparing the gas. When admitted into a jar of oxygen gas, the bursting of each bubble is attended with a brilliant flash of light and a slight concussion. The experiment should be made with caution, and the bubbles admitted singly. M. Paul Thenard has shown that this property of spontaneous combustibility is caused by the presence of the vapor of the liquid phosphide, PH2, which is always formed when the gas is procured by this process. It may be separated by passing the gas through a tube surrounded by a freezing mixture, when it condenses as a colorless liquid of high refractive power and great volatility. This body is removed also when phosphuretted hydrogen is kept for some time over water, whereby it loses its property of spontaneous inflammability. The vapor of the liquid phosphide in exceedingly small quantities will impart the property of spontaneous inflammability to pure phosphuretted hydrogen and to other combustible gases.
By the action of light it is decomposed into gaseous phosphuretted hydrogen and the solid phosphide,. P2H, which compound is often seen on the inside of vessels in which the gas is kept; it is instantly decomposed by strong acids. Pure gaseous phosphuretted hydrogen becomes spontaneously inflammable when raised to the temperature of boiling water. When the vapor of trichloride or pentachloride of phosphorus is passed over heated sal-ammoniac a compound is obtained, which was at first supposed to be dinitride of phosphorus, PN2; but according to Gerhardt it contains hydrogen, having the hypothetical formula HPN2, and has been called by him phospham. "When it is heated in hydrogen ammonia is formed. - With chlorine phosphorus forms a trichloride, PC13, and a pentachloride, PC15. The trichloride may be formed by passing dry chlorine gas over dry melted phosphorus in a retort, the vapor which distils over being collected in a receiver surrounded by ice. It is a very volatile, fuming liquid, dissolving phosphorus, and being soluble in benzole and bisulphide of carbon. It is immediately decomposed by large excess of water into phosphorous and hydrochloric acids.
The pentachloride is obtained by exhausting the air from a flask containing dry phosphorus and admitting chlorine, or by adding excess of chlorine to the trichloride. An oxychloride is formed, together with hydrochloric acid, when pentachloride of phosphorus is treated with a quantity of water insufficient to convert it into phosphoric acid. A sulphochloride of similar composition also exists, and an oxybromide and a sulphobromide, also of similar composition, and similarly obtained. There are two iodides, PI2 and PI3. Both are obtained by dissolving phosphorus and iodine together in bisulphide of carbon, and cooling the solution till crystals are deposited. There are six sulphides of phosphorus: hemisulphide, P4S; monosulphide, P2S; sesquisulphide, P4S3; tri-sulphide, P2S3; pentasulphide, P2S5; and dode-casulphide, P2S12. The mono-, tri-, and penta-sulphides unite with metallic sulphides, forming sulphur salts, the copper salts being hypo-sulphosphite of copper, CuSP2S, sulphophos-phite of copper, CuSP2S3, and sulphophos-phate of copper, CuSP2S5. Selenides are also formed of somewhat analogous composition.
Most of the metallic phosphides will be found in the articles on the respective metals. (See also Phosphor Bronze.) - Phosphorus is chiefly used in the manufacture of lucifer matches, but it is also an important article of the materia medica. In small doses it acts as a powerful general stimulant, in large doses as a violent irritant poison. When not oxidized in the stomach, it is absorbed into the system, probably dissolved in oily matter. Its action, aside from its furnishing through assimilation material to the nervous system, is directed to the kidneys and genital organs, producing diuresis, and exciting the venereal passion. It has been recommended in impotency, typhoid and typhus fevers, paralysis, locomotor ataxy, and general prostration. The usual form of exhibiting it is in oily solution, sometimes dissolved in oil of turpentine. The solution may be formed into pills, each pill containing one twentieth of a grain, and one or two of them may be taken two or three times a day. It is also given in the form of diluted phosphoric acid, when it has more the properties of a general tonic and refrigerant. The dose is 20 drops of the diluted acid, taken in a wine glass of water.
Phosphorus is also administered in the form of the hypophosphites, such as lime, soda, and potassa, and of iron, and also combined with' iron in the form of pyrophosphate of iron. The alkaline and earthy hypophosphites have obtained considerable celebrity as remedies in tubercular consumption, in combination with a generous and fatty diet; and the pyrophosphate of iron in conjunction with calisaya bark, in the form of an elixir, is often used with great benefit as a tonic.
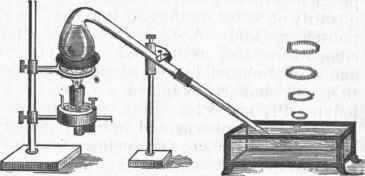
Fig. 3. - Phosphuretted Hydrogen.
Continue to:


