Potassium
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Potassium
Potassium, one of the alkali metals, discovered by Sir Humphry Davy in 1807. It had long been suspected that the alkalies and the earths were compound bodies, but up to this time all attempts to decompose them had failed. When potash was shown to be an oxide of a metal, and the metal was separated, the decomposition of the other alkalies and earths soon followed. Davy obtained potassium by subjecting a piece of moistened potassium hydroxide to the action of a powerful galvanic battery (200 Wollaston's pairs 6 in. square), the alkali being placed between a pair of platinum plates as electrodes. Gay-Lussac and Thenard in 1808 obtained it by a purely chemical process. They caused caustic potash to pass over iron turnings heated to whiteness in a gun barrel covered with clay luting to protect it from the action of the air. The oxygen of the potash combined with the iron filings, setting free the hydrogen and potassium, the latter being condensed in a well cooled copper receiver. A more recent method, invented by Ouraudau and improved by Brauner, and still further by Maresca and Donne, is the one now in general use. Many precautions are necessary to insure a good result.
The best material from which to obtain it is some potassium salt of a vegetable acid, the crude tartar of commerce (hydric-potassic tartrate) being usually employed. About 6 lbs. of this salt is ignited in a covered iron crucible till it ceases to emit combustible vapors, by which is produced a porous mass of potassic carbonate mingled with finely divided carbon. This is rapidly cooled by the application of cold water to the exterior of the crucible, and the charred mass is broken into lumps about the size of a hazel nut and quickly introduced into a wrought-iron retort, a, fig. 1, which is placed in a horizontal position in a furnace. A wrought-iron tube, 5, about 4 in. long, conveys the vapors of potassium produced during distillation into a receiver, c, which is in the form of a shallow iron box, shown on a larger scale at d, fig. 2. This box is about 12 in. long, 5 in. wide, and 1/4 in. deep between the plates, which are about 1/6 in. thick, and is open at both ends, the socket fitting upon the, tube b. The flattened form of the receiver causes a rapid cooling of the potassium and its withdrawal from the action of the carbonic oxide which is evolved during the whole process, and has a tendency to form a dangerously explosive compound.
Before adjusting the receiver the retort is slowly raised to a dull red heat, when powdered vitrified borax is sprinkled over it, forming a coating which protects it from oxidation. The heat is then increased to a reddish white, and when vapors of potassium appear and burn with a bright flame the receiver is adjusted to the tube b, which must not be exposed more than a quarter of an inch outside of the furnace, to avoid its being partially cooled, and the consequent formation of the explosive compound or the obstruction of the tube by solid potassium. The receiver is kept cool by a wet cloth fed with a cold water drip. If any obstruction forms, it must be immediately removed by thrusting an iron rod through the receiver and into the tube b. Failing in this, the fire should be immediately withdrawn. When the operation is completed, the receiver containing the potassium is detached and plunged into a vessel of rectified Persian naphtha, having a cover and kept cool by immersion in water. When cooled, the receiver may be taken apart, and the potassium detached and preserved under naphtha. When required absolutely pure, it must be redistilled in an iron retort from which the air has been expelled by the introduction of naphtha vapor.
The purified metal usually amounts to about two thirds of the cruder material first taken from the receiver. - Potassium is a brilliant bluish white metal, having a specific gravity of 0.865, being the lightest of all the metals except lithium. Its symbol is K (Jcalium), and its atomic weight 39.1. It is monatomic, belonging to the group which includes the other alkali metals, caesium, rubi-dinum, lithium, and sodium, together with silver. At 32° F. it is brittle, having a crystalline fracture, and at a little higher temperature it becomes malleable. At 59° it may be easily moulded; at a few degrees higher it becomes pasty, and at 144.5° it is liquid. When soft its clean surfaces may be welded together like iron, and at a red heat it distils as a beautiful green vapor. If exposed to the air at ordinary temperatures, it rapidly becomes covered with a film of oxide; and when raised to the point of volatilization it bursts into a bright violet flame. In contact with water, upon which it floats, it combines with its oxygen so powerfully as to produce sufficient heat to inflame the liberated hydrogen. The melted globule spins around upon the surface of the water, emitting a violet flame caused by the admixture of a small quantity of volatilized potassium with the hydrogen gas.
A film of vapor is formed between it and the water, and the decomposition of this vapor supplies the oxygen to the burning potassium. When the melted globule of potassic hydrate formed by the combustion becomes sufficiently cooled to come into contact with the water, it is scattered with an explosive burst of steam. Potassium decomposes nearly all the gases containing oxygen when heated in contact with them, and at a high temperature will remove that element from almost all bodies containing it. At a heat a little below redness it absorbs hydrogen and is converted into a grayish hydride, from which a higher heat expels the hydrogen. It forms alloys with most other metals, the combination being generally effected by fusion. Antimonide, arsenide, and bismuthide may also be formed by heating the metals with cream of tartar. - Potassium is very widely diffused in the mineral, vegetable, and animal kingdoms. It occurs as a silicate in several minerals, particularly in feldspar (orthoclase) and mica, and is therefore an important constituent of most granitic rocks. As a sulphate it is combined with sulphate of alumina in alum stone; as chloride, bromide, and iodide, in sea water and salt deposits; and as nitrate, in various soils in tropical countries.
It enters largely into the composition of the tissues and juices of land plants, especially of the grape, the apple, and other fruits, and of esculent vegetables, particularly the potato. The ashes of most forest trees abound with it. In marine plants, and in several land plants growing near the sea, the potassium is replaced by sodium; and in animals, although the tissues and fluids contain more or less potassium salts, the alkaline constituent is chiefly sodium, mostly in the form of chloride or common salt. - There are three well defined oxides of potassium: a basic or dipotassic oxide (the potash of the chemist, K20), which furnishes the salts of the alkali, a deutoxide (K202), and a peroxide (K204). The two latter do not form corresponding salts with acids. The dipotassic oxide is formed by allowing thin slices of metallic potassium to become oxidized in air perfectly free from moisture or carbonic acid, or by heating potassium with an equivalent quantity of hydrate (hydroxide), KHO + K=K20 + 11. When moistened it combines with water, with incandescence, being reconverted into hydrate. Potassic hydrate, hydroxide of potassium, or caustic potash, known in commerce as potash, is described in the preceding article.
When metallic potassium is gently heated in a current of dry air, it absorbs more oxygen than at ordinary temperatures, and the peroxide is formed; and the deutoxide is formed at a certain stage of the process. - Salts. The salts of potassium are numerous and important. 1. Chloride of potassium, KC1 (digestive salt, sal febrifugum Sylvii), may be formed by the spontaneous combustion of potassium in chlorine gas; also by passing chlorine over red-hot hydrate or iodide of potassium, by gently heating potassium in hydrochloric acid gas, by dissolving hydrate or carbonate of potash in aqueous hydrochloric acid, and in other ways. It occurs native, sometimes pure, as in sylvine, which is found in cubic crystals about the fumaroles of Vesuvius and in thin layers in the salt beds of Stassfurt near Magdeburg; but it is more frequently found mixed or in combination with other chlorides, in mineral waters, in kelp, and in mineral deposits. At Stassfurt it occurs not only as sylvine, but also as carnallite, a hy-drated double chloride of magnesium and potassium (KCl,MgCl2+6H2O), named from its pink color, in a bed of clay lying immediately above a bed of rock salt; and it is also found in the rock salt formation of Maman in Persia. Its position with regard to the rock salt is exactly that which would result from the gradual drying up of an inland sea, the common salt, from being less soluble, crystallizing out first.
Chloride of potassium crystallizes in cubes, rarely in octahedrons, composed in 100 parts of potassium 52.41 and chlorine 47.59. Its specific gravity is 1.994. It has the taste of common salt, decrepitates when heated, melts at a low red heat, and at a higher temperature volatilizes unchanged. It is sparingly soluble in alcohol. 2. Bromide of potassium, KBr. Potassium unites directly with bromine, with ignition and detonation. The salt may also be prepared by neutralizing hy-drobromic acid with potash, by decomposing bromide of iron with carbonate of potash, or by adding pure bromine to caustic potash till the liquid acquires a slight yellow color, a bro-mate being also formed, which may be decomposed by a current of sulphuretted hydrogen gas. The excess of gas is expelled by a gentle heat, and the crystals of bromide obtained by evaporation. It crystallizes in brilliant cubes, which are sometimes elongated into prisms or flattened to plates, composed in 100 parts of potassium 32.83 and bromine 67.17; specific gravity 2.672. It has a sharp taste, decrepitates by heat, and melts without decomposition. It is very soluble in water, more so in hot than cold, and is slightly soluble in alcohol. At a red heat it is decomposed by chlorine.
Bromide of potassium is an important article of the materia medica. It has of late years been largely used as a remedy for wakefulness, on account of its sedative action on the nervous system. In large doses, from 30 to 60 grains several times a day, it produces a peculiar intoxication, with torpor, drowsiness, and slowness of the pulse. Its reputed antaphrodisiac properties have been confirmed by Theilmann of St. Petersburg, Trousseau of Paris, Dr. Garrod, and others. It has been used with advantage by Sir Charles Locock in hysterical epilepsy and other nervous affections connected with uterine disorder. 3. Iodide of potassium, KI, may be prepared by several different methods, similar to those for preparing the bromide, but the best is that recommended by Liebig, which consists in decomposing iodide of barium by sulphate of potassium. The iodide of barium is prepared as follows: to 1 part of amorphous phosphorus and 40 parts of warm water there is gradually added 20 parts of dry iodine, which is triturated with the phosphorus under the water. The resulting dark brown liquid is heated on a water bath till it becomes slightly alkaline.
Insoluble phosphate and soluble iodide of barium are formed, and being separated by filtration, the filtrate containing the iodide of barium is treated with sulphate of potassium. Double decomposition ensues, by which insoluble sulphate of baryta (heavy spar) and soluble iodide of potassium are formed. The iodide may then be obtained by filtration and evaporation. It crystallizes in cubes, which are translucent or milk-white and opaque, composed in 100 parts of potassium 23.54 and iodine 76.46. Sometimes it crystallizes in octahedrons. Its specific gravity is 3.056. It is not deliquescent in a moderately dry atmosphere, is very soluble in water, and in six parts of alcohol of specific gravity 0.83. It has a sharp, bitter taste, turns reddened litmus paper slightly blue, melts below a red heat, and volatilizes undecomposed at a moderate red heat. It is decomposed by chlorine with the aid of heat into chloride of potassium and iodine. An excess of chlorine causes the formation of a double salt of terchloride of iodine and chloride of potassium, which colors the liquid yellow and yields crystals by evaporation. Iodide of potassium is one of the most important agents of the materia medica.
Its peculiar medicinal properties are principally conferred upon it by its iodine constituent. (See Iodine.) Its principal use is in reducing scrofulous and other tumors, in the treatment of skin diseases, of rheumatism, and of constitutional diseases, and in eliminating mineral poisons, particularly mercury and lead, from the system. Free iodine dissolves in a solution of iodide of potassium, forming, in the proportion of one part of iodine to two of the salt, Lugol's solution. 5. Phosphide of potassium, KP, is formed when phosphorus and potassium are heated together in hydrogen gas, the combination being attended with evolution of light and heat. It is decomposed by water, yielding' hypophosphite of potassium, phosphuretted hydrogen gas, and solid phosphide of hydrogen. 6. Fluoride of potassium, KF, is formed by dissolving potash or its carbonate in hydrofluoric acid, evaporating, and heating strongly to expel the acid. It is deliquescent, soluble in water, and crystallizes in colorless cubes, often lengthened into prisms. It forms definite crystallizable compounds with many other fluorides.
The fluoride of potassium and hydrogen, KHF2, has been recommended by Prof. Wolcott Gibbs as a convenient reagent for decomposing refractory silicates and other minerals. 7. Potassium unites with sulphur in five different proportions, forming five sulphides, K2S, K2S2, K2S3, K2S4, and K2S5. They all have an alkaline reaction to test paper, and an odor of sulphuretted hydrogen. Liver of sulphur (hepar sulphuris), prepared by heating sulphur with carbonate of potash in a- covered crucible, .is composed of a trisulphide, a pentasulphide, and some of the intermediate sulphides, mixed with sulphate, and often with carbonate of potash. It is a local irritant, and is used in the form of the unguentum potassm sulphuratm of the British pharmacopoeia. 8. There are two carbonates of potassium, a normal or neutral carbonate, or dipotassic carbonate, K2CO3, and an acid salt, monopotassic carbonate, KHCO3, commonly called bicarbonate of potash. The normal carbonate is prepared as follows. Commercial pearlash, known also as crude carbonate of potash (see Potash), which consists principally of carbonate, is agitated with an equal weight of cold water. The carbonate, being much more soluble than the other salts, is dissolved out.
The solution is then boiled down to a small bulk and left to cool, when the carbonate separates in small crystals. A purer salt may be obtained by igniting cream of tartar in a crucible, which produces a mixture of dipotassic carbonate and charcoal (black flux). The salt is obtained by digestion in water, filtration, and evaporation. It is very soluble in water, one part of the anhydrous salt dissolving in 1.05 part of water at 87.4° F. and in 0.49 part at 158°. It contains in 100 parts 68.17 of dipotassic oxide, and 31.83 of carbonic acid; specific gravity 2.267. It crystallizes from an aqueous solution in oblique rhombic octahedral crystals, containing two equivalents of water (K2CO3 + 2H2O), which are very deliquescent. Its reaction with test paper is strongly alkaline, and it has an acrid alkaline taste. The anhydrous salt fuses at a red heat and is partially volatilized at a high temperature. When it is heated to redness with silica, the latter unites with the oxide, forming silicate of potassium; and advantage is taken of this affinity in the analysis of mineral substances containing much silica.
Dipotassic carbonate is extensively used in the arts, as in the manufacture of soap and glass, and in preparing caustic potash and other chemical compounds for the purposes of pharmacy and chemistry. The monopotassic carbonate, commonly called bicarbonate of potash, is obtained by passing carbonic acid gas into a solution of the normal carbonate in five parts of water. The bicarbonate, being much less soluble than the normal salt, is precipitated, and being collected on a filter, pressed, and redissolved in warm water, crystallizes on cooling in large rhomboidal prisms belonging to the monoclinic system, which are soluble in four parts of cold and five sixths their weight of boiling water, and are nearly insoluble in alcohol. Bicarbonate of potash is much used in chemistry when a pure potassium salt is required, and is an important article of the pharmacopoeia. Its medicinal properties are similar to those of the carbonate,-and being milder in its action it is generally preferred. It is used as an antacid in some forms of dyspepsia, in acute rheumatism, for which it is a standard remedy, and also in many cutaneous affections.
It increases the alkalinity of the blood, and facilitates glandular secretion, for which reason it is beneficial in most forms of fever and inflammatory diseases. 9. Nitrate of potassium (nitre, saltpetre) is described in the article Nitrates. 10. The principal sulphates of potassium are a normal or neutral sulphate and an acid sulphate. Normal or bipotassic sulphate, K2SO4 (nitrum mtriolatum, tartarus vitriolatits, specificum purgans Paracelsi), occurs native in delicate needle-shaped crystals, or as a crust on many Vesuvian lavas, and in this form is often called glaserite, arcanite, aphthalose, and Vesuvian salt. It exists also in solution in sea and mineral waters, and in vegetable and animal fluids. It is obtained as a by product in several manufacturing processes, as in the manufacture of nitric acid from saltpetre, where the residue, consisting principally of acid sulphate, is neutralized with crude potassic carbonate. The hot solution on cooling yields crystals of normal sulphate, either in four-sided oblique rhombic prisms, or in six-sided pyramids, belonging to the trimetric system.
They are anhydrous, require about 12 parts of water at 32° F. for solution, and are composed in 100 parts of dipotassic oxide 54.07, and sulphuric anhydride 45.93; specific gravity 2.66. They'are insoluble in alcohol, and decrepitate strongly when heated. Sulphate of potash is a mild purgative, but is used in medicine principally as an ingredient of Dover's powder, or pulvis ipecacuanha compositns. The acid, or monopotassic sulphate, KHS04, commonly called bisulphate of potash, is formed by mixing the neutral sulphate with half its weight of oil of vitriol, evaporating to dryness in a platinum vessel, and dissolving the fused salt in hot water, from which it crystallizes on cooling, in flattened rhombic prisms, which dissolve in two parts of water at 60° F., and in less than one part of boiling water. It occasionally crystallizes in anhydrous needles having the formula K2S04,S03. 11. Chlorate of potassium, or potassic chlorate, commonly called chlorate of potash, is analogous in composition to chloric acid (HClO3), the difference being in the substitution of an equivalent of potassium for one of hydrogen. "When chlorine gas is passed into a hot solution of potash or carbonate of potash, the liquid yields on cooling crystals of chlorate of potassium, KC103. The salt is made in this indirect way instead of adding chloric acid to potash, and precedes the formation of the acid, which is obtained by boiling the salt in a solution of hydrofluosilicic acid.
It is a simple example of chemical substitution, a subject the study of which has thrown much light upon the science of chemistry. Chlorate of potash is prepared in the large way by converting milk of lime into, a mixture of calcic chlorate and chloride with an excess of chlorine, and adding potassic chloride, by which the calcic chlorate is decomposed (Ca2C103+ 2KCl=2KC103 + CaCl2). The potassic chlorate, being sparingly soluble, is easily separated from the very soluble calcic chloride by evaporation, and deposited in six-sided prisms, which being redissolved in boiling water again crystallize in six-sided plates belonging to the mono-clinic system. They dissolve'in about 16 parts of water at 60° F., and in about 1.6 part of boiling water. When heated, the salt gives off the whole of its oxygen, chloride of potassium remaining; a perchlorate is formed in the early stages of the decomposition. Chlorate of potash is a powerful oxidizing agent, and detonates violently when mixed with certain organic bodies and heated or struck with a hammer. If mingled with flowers of sulphur and triturated in a mortar, it produces a series of sharp detonations. It is used in the manufacture of lucifer matches, in percussion caps, and in the composition of fireworks.
The following composition is applied to the interior of percussion caps: chlorate of potash 26, nitre 30, fulminate of mercury 12, sulphur 17, ground glass 14, gum 1 = 100 parts. Chlorate of potash is extensively used as an oxidizing agent in heightening the intensity of steam colors on printed goods, and in the chemical laboratory as a source of oxygen. (See Oxygen.) It is not adapted to the manufacture of gunpowder, as it is too violently explosive, endangering the bursting of the gun, and is also liable to become ignited by friction. (See Explosives.) It is one of the most valuable agents of the materia medica, and is employed in the treatment of scurvy, rheumatism, pseudo-membranous diseases, as croup, diphtheria, and scarlet fever, and as a prophylactic in mercurial salivation. Internally it may be given in doses of from 5 to 30 grains three or four times a day, but is generally used in much smaller quantities; as a gargle in diphtheria, from half an ounce to an ounce may be dissolved in a pint of water. 12. Several salts (phosphates of potassium) result from the union of potassium with the different forms of phosphoric acid (see Phosphorus), which are not of sufficient general interest to warrant a description. - Potassium forms important salts in combination with cyanogen (ON), and also with this radical and other elements. 13. Cyanide of potassium, KCN or KCy, is formed by burning potassium in cyanogen gas, and also when the vapor of hydrocyanic acid is passed into a cold alcoholic solution of potash; but it is usually prepared from ferrocyanide of potassium.
Eight parts of this salt are mixed with three of carbonate of potash and heated to redness in a covered iron crucible. The iron constituent separates in the form of a metallic powder and settles to the bottom of the crucible, and the fused cyanide being poured off solidifies to a milk-white mass. Potassium cyanide is also produced in considerable quantities in blast furnaces in which ores are smelted with coal or coke. It crystallizes in anhydrous cubes or octahedrons, which deliquesce in the air and are very soluble in water. It is a powerful reducing agent; the oxides of many of the metals, when thrown into the melted salt, are readily reduced to the metallic state, cya-nate of potassium being formed. On account of this property it is useful in removing stains of metallic oxides, as indelible ink and the juices of fruits. With many of the metallic salts it forms precipitates of cyanides, which are generally soluble in excess of potassic cyanide, forming crystallizable double salts. Thus, when it is added to a solution of nitrate of silver, cyanide of silver is precipitated, which being washed and treated with another portion of the potassic cyanide, there is formed a solution of a double cyanide of silver and potassium, AgKCy2. A similar solution of gold and of other metals may be formed in the same way, and these are in common use in electroplating. (See Galvanism, vol. vii., p. 600.) 14. Ferrocyanide of potassium, potassic ferrocyanide, or yellow prussiate of potash, is an important salt, met with in commerce nearly pure, and is the source from which the cyanogen compounds are usually obtained.
It may be formed by digesting ferrous hydrate in a solution of potassic cyanide; but it is made on a large scale by heating azotized matter to redness with potassic carbonate and iron. Dried refuse animal matters, as blood, horns, and parings of hides, are mixed with an equal weight of crude commercial potash, which contains sulphate of potash and about one third as much iron filings, and heated to redness in a large iron retort from which the air is excluded. The melted mass is then treated with hot water, which completes the chemical combination. The reactions by which the salt is produced are complicated. Cyanogen is first formed by the reaction of the potash and iron on the azotized matter, and the cyanogen then combines with the potassium, forming potassic cyanide. A quantity of iron is at the same time converted into sulphide from the action of potassic sulphide, which is produced by the action of the sulphate of potash contained in the crude potash. When the mass is treated with water the potassic cyanide attacks both the metallic iron and the sulphide of iron, forming potassic ferrocyanide, which is obtained from the filtered liquor by evaporation in large transparent lemon-yellow crystals, derived from an octahedron with a square base, with easy cleavage parallel to the base, having the formula K4FeCy6 + 3H20. They dissolve in four parts of cold and in two parts of boiling water, but are insoluble in alcohol.
Exposed to a gentle heat, the three equivalents of water are expelled. Raised in a closed crucible to a high heat, potassic cyanide, iron carbide, nitrogen, and other gaseous products are formed. Potassic ferrocyanide is one of the most valuable of chemical reagents. With neutral or slightly acid salts of the heavy metals it forms precipitates having characteristic colors, the potassium being generally displaced by the new metal. It is not poisonous, and is sometimes used in medicine as a sedative tonic. When it is distilled with sulphuric acid, an abundance of hydrocyanic (prussic) acid is produced. It is largely used in dyeing and calico printing in connection with certain mordants upon which the color produced depends. It is used in the preparation of Prussian blue, which is the dye formed when cloth mordanted with an iron salt is treated with a solution of potassic ferrocyanide. With ferrous salts (proto-salts of iron) potassic ferrocyanide produces a white precipitate of potassic-ferrous ferrocyanide (K2Fe2Cy6), which on exposure to the air is converted into Prussian blue or ferrocyanide of iron.
Prussian blue is more directly produced by adding potassic ferrocyanide to a ferric salt (per-salt or iron); a deep blue precipitate is immediately formed, consisting of ferrocyanide (Fe7Cy18), which in combination with 18 molecules of water constitutes Prussian blue. The Prussian blue of commerce is, however, prepared from the ferrous salt, the white precipitate being allowed to turn blue by exposure to the air or the action of nitric acid. The product is not pure ferric ferrocyanide, for a reaction takes place by which a potassic-ferrous ferricyanide is formed, which also has a beautiful deep blue color. When freshly precipitated Prussian blue is very bulky, but in drying it shrinks to a hard and brittle mass resembling indigo in appearance. It is quite insoluble in water and dilute acids, with the exception of oxalic acid, with which it forms a deep blue liquid, sometimes used as an ink. It is decomposed by alkalies with loss of color, the alkali combining with the ferrocyanide, ferric oxide being separated. Heated in the air it burns like tinder, leaving a residue of ferric oxide. It forms a beautiful pigment as an oil or water color, but has little permanence. It is used in medicine as a tonic, febrifuge, and alterative.
It has been recommended by Dr. Zollickoffer of Maryland in intermittent and remittent fevers, as being more prompt and efficacious than quinia; and has been given by Dr. Kirchoff of Ghent with advantage in epilepsy, and by Dr. Bridges of Philadelphia in severe and protracted facial neuralgia. The dose is from three to five grains four or five times a day. 15. Ferricyanide of potassium, potassic ferricyanide, or red prussiate of potash, is an important reagent of the chemical laboratory and the calico printer. It is prepared by slowly passing chlorine gas through a dilute cool solution of potassic ferrocyanide until the liquid acquires a deep red color and ceases to precipitate a ferric salt. The reaction may be represented as follows: 2K4FeCy6 + Cl2=2K3FeCy6 + 2KCl, the chlorine withdrawing one fourth of potassium from the yellow salt. It is separated from the potassic chloride by evaporation and crystallization, and is obtained pure by re-crystallization in the form of transparent, anhydrous, right rhombic prisms, of a beautiful ruby red, often large. They are soluble in four parts of cold and one and a third part of boiling water; insoluble in alcohol. The salt may also be obtained by acting on the yellow prussiate by various oxidizing agents.
Potassic ferricyanide, when added to a ferric salt, produces no precipitate; but with a ferrous salt it produces a deep blue precipitate of ferrous ferricyanide, which is known as Turn-bull's blue. It is therefore a delicate test for a ferrous salt. - There are several organic salts of potassium, the principal of which are the tartrates, acetates, oxalates, and citrates. 16. There are two tartrates, a normal and an acid salt. The normal salt, normal potassic tartrate, or soluble tartar (K2C4H406), is prepared by neutralizing the acid salt (cream of tartar) with chalk or carbonate of potash. It is very soluble, and crystallizes with difficulty in right rhombic prisms, which are permanent in the air and have a bitter saline taste. It is used in medicine as a mild cooling purgative, sometimes combined with senna. The dose is from a drachm to an ounce. The acid salt, hydric-potassic tartrate or bitartrate of potassium (KHC4H4O6), is described under Cream of Tartar. A double tartrate of potassium and sodium is the beautiful Rochelle or Sei-gnette salt. (See Rochelle Salt.) 17. There are two acetates of potassium, a normal and an acid salt. The normal salt, normal potassic acetate (arcanum tartare, sal diureticvs), exists in the juices of many plants.
It may be prepared by neutralizing acetic acid with potassium carbonate and heating the salt to fusion. Its formula is KC2H3O2. It may be obtained by the double decomposition of potassic sulphate and calcic acetate. It is a white salt, of a pungent saline taste, perfectly neutral to test paper, extremely deliquescent, becoming converted into a liquid of an oily appearance on exposure to the air; it must therefore be kept in well stoppered bottles. It dissolves in about half its weight of water, and in twice its weight of alcohol. Heated above its point of fusion, it is decomposed into acetone and potassic carbonate. It is an important article of the materia medica, acting as a diuretic in doses of from 20 to 60 grains, and as a mild cathartic in two or three drachm doses. Dr. Eaton of Glasgow has found it useful in several skin diseases, as eczema and lepra; and the late Dr. Golding Bird found it remarkably efficacious in the treatment of acute rheumatism, the pain of the disease declining as soon as the urine became alkaline. The acid salt, diacetate of potassium, or acid potassic acetate, KC2H3O2, C2H4O2, is prepared by adding to a solution of the neutral salt an excess of acetic acid. By slow evaporation the salt crystallizes out in long flattened prisms.
It is very deliquescent, melts at 298° F., and parts with one equivalent of acetic acid in a crystalline form at 392°. 18. There are two oxalates. The neutral salt, K2C2O4 + 2H2O, prepared by neutralizing oxalic acid with potassic carbonate, crystallizes in transparent rhombic prisms, which become opaque and anhydrous by heat, and dissolve in three parts of water. The acid oxalate, or binoxalate, KC2HO4 + 2H2O, called also salt of sorrel, occurs in various species of rumex, as R. acetosa or common sorrel, in oxalis acetosella (wood sorrel), and in garden rhubarb, associated with malic acid. It is prepared by dividing a solution of oxalic acid into two equal parts, neutralizing one with potassic carbonate, and adding the other. The salt crystallizes on cooling in colorless rhombic prisms, soluble in 40 parts of cold and in 6 parts of boiling water, and has an acid reaction. A solution of salt of sorrel is often used for removing ink and fruit stains from cloth and paper. Its efficacy in removing ink depends on the solubility of the double salt, oxalate of iron and potassium, which is produced. 19. There are three citrates, tripotas-sic, dipotassic, and monopotassic citrate.
Tri-potassic citrate, citrate of potassium, or common citrate of potash, K3C6H5O7+ H20, is formed when a solution of citric acid is neutralized by potassic carbonate. On evaporation it crystallizes in transparent stellate-grouped needles, which lose their water of crystallization at 392° F. They are very deliquescent, and insoluble in alcohol. Citrate of potash is adopted as a refrigerant and diaphoretio in the United States and British pharmacopoeias, and is sometimes prepared extemporaneously as an effervescent draught. Dipotassic citrate, K2C6H6O7, made by adding an equivalent of citric acid to the tripotassic salt and evaporating, and the monopotassic citrate, KC6H707, are both acid salts, the latter crystallizing with 13.8 per cent, of water of crystallization and having an agreeable sour taste. Malate of potassium exists to a certain extent in the juice of some fruits, but malic acid more commonly exists in combination with calcium.
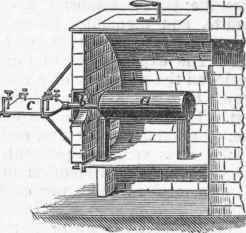
Fig. 1. - Potassium Furnace.
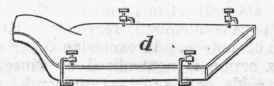
Fig. 2. - Receiver.
Continue to:


