Caoutchouc, Or India Rubber
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Caoutchouc, Or India Rubber
Caoutchouc, Or India Rubber (called by the South American Indians cahuchu), the inspissated milky juice of a number of trees and plants found in Mexico and Central America, in Brazil, Guiana, Peru, and in the East Indies. The Mexican tree is the castilloa elastica, a genus belonging to the order artocarpacem. The South American tree has been variously named by different naturalists. It is the jatropha elastica of Linnaeus, the siphonia elastica of Persoon, the siphonia cahuchu of. Scherber and Willdenow, and the hevea Ouianensis of Au-blet, and belongs to the order euphorliacem.

Canvas-Back Duck (Aythya Vallisneria).
The trees which furnish the greater part of the caoutchouc brought from the East Indies are the Jicus elastica of Assam, of the order mora-cea, and the urceola elastica of Sumatra and other islands of the eastern archipelago, which belongs to the order apocynacea. The ficus elastica is one of the noblest of trees, and beyond the Ganges are found inexhaustible forests of it. Several years ago William Griffith, an Englishman, published a report upcn this caoutchouc tree of Assam at the request of Capt. Jenkins, the agent of the governor general of India, in which he says the tree is either solitary, or in two- or three-fold groups; is large and umbrageous, and may be distinguished from other trees at a distance of several miles by the picturesque appearance of its dense and lofty crown. The main trunk of one measured 74 ft. in circumference, and the area covered by its expanded branches had a circumference of 610 ft. The height of the central tree was 100 ft. It has been estimated that there are over 43,000 such trees in the district of Chard-war, in an area 30 m. in length by 8 in width. It grows on the slopes of the mountains from the valleys up to an elevation of 22,000 ft.
Its geographical range in Assam appears to be between lat. 25° and 27° N, and between about Ion. 90° and 95° E. It is allied to the banian (ficus Indica). The urceola elastica, a twig of which in blossom, with some of the fruit, is represented in fig. 2, is a large climbing tree, with a trunk frequently as thick as a man's body. It has sharp, ovate-oblong, opposite leaves, with a rough upper and hairy under surface, and bears many-flowered terminal cymes of small greenish blossoms, which produce double fruits, consisting of two large, roundish, .apricot-colored, rough, leathery-skinned pieces, about the size of oranges, containing numerous kidney-shaped seeds in a copious tawny-colored pulp, which is much relished both by natives and European residents. The flowers have a five-cleft calyx; a pitcher-shaped hairy corolla, with five short, erect teeth; five stamens, rising from the base of the corolla, and having very short filaments and arrowhead-shaped anthers. The castilloa elastica, the Mexican tree, grows from 50 to 100 ft. high and from 8 to 20 in. in diameter. It has male and female flowers alternating on the same branch. The male flowers have several stamens inserted into a hemispherical perianth, consisting of several united scales.
The female flowers consist of numerous ovaries in a similar cup. The South American tree, the siplionia elastica, varies from 25 to upward of 100 ft. in height. The leaves consist of three entire leaflets radiating from the top of a long stalk, and are clustered toward the ends of the branches. The flowers are borne in loosely branched panicles, with numerous little branchlets consisting of a few male flowers and a female at the top. Both sexes have a bellshaped five-toothed calyx, and no corolla. The fruit is a large capsule composed of three one-seeded pieces, which split in leaves when ripe. The raw seeds are poisonous to man and quadrupeds, but macaws eat them, and they are used as a bait for fish. Long boiling deprives them of their poison. - The province of Para, south of the equator, in Brazil, furnishes to commerce immense quantities of caoutchouc. The tree is tapped in the morning, and during the day a gill of fluid is received in a clay cup placed at each incision in the trunk. This when full is turned into a jar, and is ready at once to be poured over any pattern of clay, or a wooden last covered with clay, the form of which it takes as successive layers are thus applied.
Its drying and hardening are hastened by exposure to the smoke and heat of a fire, and thus the substance acquires its ordinary black color. Dried by the sun alone, it is white within and yellowish brown without; when pure, it is nearly colorless. Complete drying requires several days' exposure to the sun; during this time the substance is soft enough to receive impressions from a stick, and is thus ornamented with various designs. The natives collect it upon balls of clay in the forms of bottles and various fanciful figures, in which shapes it is often exported. They also make it into tubes, which they use for torches. The clay mould over which the bottles are formed, being broken up, is extracted through the open neck. From the custom among the natives of presenting their guests with one of these bottles furnished with a hollow stem, to be used as a syringe after meals for squirting water into the mouth, the Portuguese gave the name of seringat, or syringe, to the gum and also to the tree which produces it. The moulded articles are brought into Para suspended on poles to keep them from touching each other, as for a long time they continue sticky.
It is not only prepared in various moulded forms, as bottles, toys representing animals, rudely shaped shoes, and in flat cakes also for exportation, but a method has been devised for preserving the juice as it comes from the tree, and shipping this in air-tight vessels of tin or glass. The liquor is first filtered and mixed and well shaken with about 1/18 of its weight of strong ammonia. On being poured out upon any smooth surface, and exposed to a temperature of 70° to 100° F., the ammonia, which preserved it from the action of the oxygen of the atmosphere, evaporates, and leaves the gum in the form of the object which holds it. It has in this state a pure white appearance. The juice is of a pale yellow, of the thickness of cream, of a sourish odor, and of specific gravity 1.012. The pure caoutchouc which separates from it, rising like coagulated albumen to the surface, as the mixture of the juice with water is heated, has the specific gravity of only 0.925. This, being skimmed off like cream as it forms, is found to constitute about 32 per cent, of the juice. It may also be precipitated by salt or hydrochloric acid. On being pressed between folds of cloth and dried, it becomes transparent.
It swells by long-continued exposure to boiling water, but regains its form after being removed some time. Alcohol does not dissolve it, but precipitates it from its solution in ether. It is slightly soluble in ether free from alcohol, more soluble in coal naphtha, oil of turpentine, and chloroform; but its best solvents are sulphide of carbon, and especially the liquid hydrocarbon, called caoutchoucine, obtained by distilling the crude caoutchouc. The weak acids and alkaline solutions have no effect upon it. On evaporating its solution, the substance is recovered in some instances elastic and dry as before, so as to serve the purposes of a varnish, which possesses the properties of the original juice; or it is obtained in an adhesive, inelastic state. At the temperature of about 248° F. it melts and remains in a sticky condition, unless long exposed to the air in thin layers. It readily inflames and burns with much smoke. Its elasticity is very remarkable; and when a piece of it is stretched, heat and electricity are evolved. If a piece is kept distended for two or three weeks, its elasticity is lost; exposure to temperature as low as 40° produces the same effect; but the application of a gentle heat immediately restores it.
This property is taken advantage of in the manufacture of elastic textile fabrics, woven of the inelastic threads, which are afterward made elastic by heat. At 600° it is partially volatilized, and the vapor when condensed is the oily substance called caoutchoucine, which has been before mentioned as the best solvent of caoutchouc. According to experiments made many years ago at Utrecht, it yields at a red heat about 30,000 cubic feet of hydrocarbon gas to the ton, quite free, of course, from sulphur and ammonia compounds, and possessing illuminating properties much superior to those of the best oil gas; but its expensiveness prevents its being used in this way. According to the analysis of Prof. Faraday, the gum is a hydrocarbon consisting of 8 equivalents of carbon and 7 of hydrogen, which would require the proportion of 87'27 of carbon and 12.73 of hydrogen in 100 parts. The numbers found by him were respectively 87.2 and 12.8. The juice, as obtained from the tree, gave in 100 parts:

Fig. 1. - Ficus elastica.

Fig. 2. - Urceola elastica.

Fig. 3. - Siphonia elastica.
Water containing a little free acid......... | 56.37 |
Caoutchouc......... | 31.70 |
Albumen........ | 1.90 |
Wax..................................... | trace. |
A nitrogenized body soluble in water........ | 7.13 |
A substance insoluble in water......... | 2.90 |
100.00 |
Caoutchouc was long known before its. most valuable qualities were appreciated. La Con-damine was the first to give a particular description of the gum, which he did in a communication to the French academy of sciences in 1736. Again in 1751, after a residence of ten years in the vafley of the Amazon, he brought the subject into notice, and called attention to the memoir of M. Fres-neau, who had found the tree in Cayenne. In 1761 MM. Herissent and Macquer reported their chemical observations on caoutchouc to the royal academy; and in 1768 M. Grossait published his experiments for obtaining good tubes of India rubber by means of ether and boiling water. Dr. Priestley refers to it, in the preface of his work on "Perspective " (1770), as a substance which had just been brought to his notice, as admirably suited for rubbing out pencil marks) and as being then sold at the rate of 3s. sterling for a cubical bit of about half an inch. In 1797 a patent was obtained in England by a Mr. Johnson for rendering cloth water-proof by covering one side with a varnish made of India rubber dissolved in equal parts of oil of turpentine and spirits of wine, and sifting over the surface silk, wool, flock, and other substances; and in 1813 a patent was issued in the United States to Jacob F. Hummel of Philadelphia for a varnish of gum elastic.
In 1819 Mr. Mackintosh, who was engaged in the manufacture of cudbear, made an arrangement with the Glasgow gas works to receive their tar and ammoniacal products. It occurred to him that the oil of naphtha obtained from these might be useful as a solvent for India rubber, and in 1823 he obtained a patent for the manufacture of water-proof fabrics, since widely known as "mackintoshes." He established a manufactory at Glasgow, and subsequently, with others, went into the business on a large scale at Manchester. Mr. T. Hancock, who became associated with him, had already in 1820 obtained a patent " for an improvement in the application of a certain material to various articles of dress, and other articles, that the same may be rendered elastic." In 1825 Thomas C. Wales, a merchant of Boston, introduced the original Para overshoe in its rough state as made by the Indians of Brazil, and soon caused an improvement in its shape by sending the native shoemakers American lasts. In 1828 nearly half a million India-rubber overshoes were exported from Brazil to Europe and the United States. In 1832 Wait Webster of New York received a patent for attaching soles to gum elastic boots and shoes, and the next year similar patents were granted to Nathaniel Ruggles of Bridgeport, Conn., and to Samuel D. Breed of Philadelphia. In 1833 boots were exhibited at the fair of the American institute by J. M. Hood of New York, which had been made here and sent to South America to be varnished with the fresh juice of the caoutchouc tree.
In 1826 and 1827 Messrs. Rattier and Guibal, proprietors of a factory at Saint-Denis near Paris, employed machinery for cutting filaments of India rubber, of which tney made fabrics. Subsequently machines for this purpose were patented in England by West-head of Manchester, Mr. Nickels, and others, which cut threads from a flattened disk of rubber varying in fineness from 700 to 5,000 yards to the pound. In April, 1831, George H. Kichards of Washington, D. C, received a patent for a fluid caoutchouc; and soon afterward Edwin M. Chaffee of Roxbury, Mass., and others established the Roxbury India-rubber company, which was chartered in 1833, and was the first company organized in the United States to manufacture caoutchouc into water-proof clothing. They made shoes, coats, life-preservers, and carriage traces, covering them with caoutchouc dissolved in some essen-tial oil, spirits of turpentine being principally used. Considerable excitement grew out of this enterprise, and shares in the company's stock, which at first sold for $100, went up to $300 and $400. Active competition soon sprung up, and during that year six companies were chartered in Massachusetts for making India-rubber goods; and these were soon followed by others in Troy, Providence, and elsewhere.
In 1834 Mr. Chaffee obtained a patent for manufacturing India-rubber hose, and another for making boots and shoes. In the same year Dr. Alexander Jones of Mobile, Ala., produced a kind of carpet by means of India-rubber varnish applied to canvas which had been covered with wall paper. In September, 1835, Charles Goodyear, after devoting much time to experiments, took out his first patent, which was for an India-rubber cement. In the following year he used nitric acid to deprive the surface of India rubber of its adhesiveness, enabling manufacturers to expose an India-rubber surface on their goods; and this improvement was used till the production of vulcanized rubber. In the summer of 1838 Mr. Goodyear became acquainted with Nathaniel Hay ward, who had been employed as foreman of the Eagle company at Woburn, Mass., where he had made use of sulphur by impregnating the solvent with it, and it was from him that he received his first knowledge of the use of this material. Mr. Goodyear soon afterward occupied the factory at Woburn, and employed Mr. Hayward in manufacturing life-preservers and other articles, by the sulphurous acid gas and solarizing process; he purchased the sulphuring process of him and took out a patent for it as his assignee, Feb. 24, 1839. The sulphur process, however, would not have been of much value but for subsequent improvements made by Mr. Goodyear. The sulphur imparted an offensive odor, and did not prevent the rubber from hardening in cold weather.
Continued experiments revealed to him the fact that the application of considerable heat would cause the sulphured article to become pliant in cold weather, to have its elasticity increased at all temperatures, and its offensive odor much diminished. The new product of vulcanized rubber was the result of these experiments, and a patent was obtained for it, June 15, 1844. This patent was reissued in 1849, extended in 1858, and again reissued in 1860. Mr. Nelson Good-year's subsequent improvements, and the production of hard rubber or ebonite by him, have left little room for further improvement in the vulcanization of caoutchouc. (See Goodyear.) Many of Mr. Goodyear's patents are for the mixing of white lead and other mineral substances with caoutchouc, partially dissolved in some essential oil, grinding them together, and subjecting them to heat; and many such substances are still used for the manufacture of various articles. But most of the physical properties for which vulcanized rubber is remarkable are obtained by a simple mixture of caoutchouc and sulphur, the latter being added in quantities varying from 5 to 50 per cent., the larger quantities of sulphur being used, with an increase of heat, for the production of hard rubber, or ebonite.
With the use of a moderate quantity of sulphur and a degree of heat varying from 220° to 275° F., the compound is remarkable for possessing a much higher degree of elasticity than pure caoutchouc, and for retaining this at temperatures even below the freezing point of water; neither is this property lost by frequent stretching. It also bears a considerable degree of heat without change, depending on the amount of sulphur with which it has been combined, and the degree of heat to which it has been subjected during the process of manufacture. Its elasticity is so much increased by vulcanization, that the original article will scarcely bear a comparison. To test its power of enduring heavy blows, Mr. Brockedon subjected a piece of it an inch and a half thick and two inches square to one of Nasmyth's steam hammers of five tons' weight. When resting upon it, the caoutchouc was compressed to about half its thickness. The hammer was then lifted two feet and dropped upon it without injury. When dropped through a space of four feet the cake was torn, but its elasticity was not destroyed. - The method of vulcanization of caoutchouc has undergone important changes since its invention, both in the preparation of the crude material and in the process of adding the sulphur.
At first the caoutchouc was cut up into small bits and shreds by knives placed on strong iron cylinders, revolving in hollow cylinders armed with teeth, after which it was immersed in melted sulphur and subjected to pressure; or it was partially dissolved in oil of turpentine and ground with sulphur and other materials; or in some cases dissolved in sulphide of carbon, and this process is still sometimes employed. The most approved method, however, and the one in general use in this country, is the following: The crude caoutchouc is placed in a large vat of water, which is kept boiling by the introduction of jets of steam. In a vat which will contain about three hogsheads, 200 or 300 lbs. of the crude material may be immersed. The clay and dirt are softened as well as the caoutchouc, which also slightly swells and increases in elasticity and pliability. A mass of it, say from 10 to 20 lbs., is then taken and thrown upon a pair of strong fluted cast-iron cvlinders, between which it is masticated into small pieces, and washed by streams of hot water which fall upon it from a perforated horizontal pipe.
After being passed several times through this machine, it is taken to another, standing beside it, similar in construction, but having a pair of smooth cylinders in place of the fluted ones. These produce an enormous pressure, which packs the pieces together in the form of a mat; this is also passed several times in succession through the machine and washed by the dripping of hot water, as in the preceding operation. These machines are called washing, masticating, and compressing machines. When the mat is sufficiently compacted and washed, it is taken to a drying room, a warm chamber heated by steam, where it is allowed to remain from four to six weeks, until it is thoroughly dry; for if it were attempted to work the material while it contained any moisture, an inferior fabric would be the result. When perfectly dry the rubber is taken to the mill room and passed successively through three mills. All the mills are of similar construction to the one already represented, except that in each machine one cylinder is made to revolve twice as rapidly as the other, in consequence of which the material is thoroughly ground and mixed.
But while undergoing the process the continuity of the mat is not destroyed, for it retains its form, although a careful scrutiny will show that a constant and rapid change of position is going on among the particles. The cylinders are hollow and are supplied with steam, which keeps them at about 220° F. in the first mill, and at a little lower temperature in the other two. The first mill merely works and compresses the material into a firm thick sheet of a homogeneous texture, preparatory to the incorporation of the sulphur and whatever other ingredients are to be added, which operation is performed entirely in the second mill. Taking as an example the manufacture of India-rubber hose for steam fire engines, as carried on at a large establishment in New York, the subsequent steps are as follows: After leaving the first mill, about 5 per cent, of sulphur (and in some cases certain mineral matters, as white lead) is thrown upon the sheet while it is passing down between the cylinders. The mixing at first causes disintegration and the separation of the material into shreds; but union is speedily reestablished, and the mass again becomes homogeneous, and will retain its pliability and elasticity after cooling.
This, however, is not allowed to take place until it is passed through the third or finishing mill. Here the sheet is passed between the cylinders over and over again, until its pliability and working qualities are perfected, and as far as possible adapted to being spread upon canvas. This operation is performed in an adjoining room upon a calender (fig. 5), a machine somewhat similar to that used in cotton-bleaching establishments. The rubber is first of all again passed through a pair of cylinders in a machine called a feeder, which is also similar to the mills through which it has already passed. This feeder stands near the calender, and its purpose is to knead and temper the India rubber to the exact condition in which it can be best applied to the cloth. It is taken in handfuls at a time and fed to the calender between the two upper cylinders represented in the figure, but upon the opposite side to that which is shown. The surfaces of the two cylinders are so prepared that the rubber adheres in a thin sheet to the lower one of the two, which in its revolution brings it in contact with the third or next lower cylinder, over which the cloth is being passed, forcing it thoroughly into the meshes of the fabric.
After one side of the canvas has been coated it is turned, and the rubber is applied to the other side. It is then taken to a larger calender, where another coating is applied to one side, the whole sheet being well consolidated under powerful pressure. The cloth is now ready to be made into hose, and the operation is commenced by cutting it into strips diagonally, so that both warp and weft may receive the strain to which the hose may be subjected, thus greatly increasing the strength of the fabric. The strips are cut in width a little more than twice the intended circumference of the hose, so that one sheet will form two thicknesses of its walls.

Fig. 4. - Washing and Compressing Mill.

Fig. 5. - Calender.
The inner layer of the pipe is formed by a thick sheet of uncanvassed vulcanized rubber, which has been also prepared in one of the calenders. This is cut of the proper width, and wound round a long iron pipe used as a mandrel, and its edges are lapped over one another, firmly pressed together, and permanently joined by a small grooved roller held in the hand of the workman. Before being applied, the inner surface of this sheet of rubber must be coated over with a powder of some substance which will prevent adhesion to the mandrel, so that it may be removed after the hose is finished. The best substance is soapstone, or steatite. The lapping edge must be carefully left untouched with this material, or perfect union will not be possible. Around this inner coating are now successively wrapped two strips of the bias-cut rubber canvas, and over this another and outer coat of pure vulcanized rubber, making six coats in all, four of which are of rubber canvas. It is claimed that hose of two inches calibre, made in this manner, is capable of resisting a hydrostatic pressure of 400 lbs. per square inch at a temperature of 60° F. Each length of hose is usually made 50 ft. long, which has been found the most convenient for use on the hose carriages, the lengths being joined as required by couplings.
After every layer has been wound over its concentric fellow, and also during the process, the workmen make use of their rollers to compress and consolidate the hose. After all the layers have been applied, the pipe is taken to another bench, where it is covered with four or five layers of cotton cloth, and then, with several others, it is placed upon a long carriage which runs upon rails into a large hollow cylinder which is heated by live steam, or steam which is not superheated, coming immediately from the boiler, and usually at a pressure which will give it a temperature of about 240° F. When the rubber has been confined in this cylinder, at this temperature, for eight or ten hours, the true vulcanization or union of the caoutchouc with the sulphur takes place, accompanied with the disengagement of sulphuretted hydrogen gas. This is one of the most important parts of the process of manufacture, and upon it, as well as upon the mixing of the ingredients, depend the strength and elasticity of the product. The heat should be raised gradually and maintained at a determined point till the vulcanization is completed, and then should be immediately withdrawn.
In manufacturing engine hose, the New York gutta percha and rubber manufacturing company mix a certain amount of carbolic acid with the caoutchouc, which it is claimed preserves the hose and shortens the process of vulcanization. An ingenious register is in use at their factory, the invention of Mr. John Murphy, by which the application of a steam pressure gauge to clockwork records the different degrees of temperature, and their duration, which may have been reached during the vulcanizing process, which is generally performed during the night, under the care of one or two men. When caoutchouc is intended for car springs, about 5 per cent, of white lead and variable proportions of carbonate of lime are added, with 5 per cent, of sulphur. This makes the product more solid and substantial, and capable of supporting greater weight without too much compression, which is objectionable. In the manufacture of ebonite, as before stated, a much larger proportion of sulphur is used; and in the cheaper kinds, when great strength is not required, various earthy substances are employed. But sulphur and caoutchouc alone, when properly mingled and raised to the required degree of heat, produce the best article.
The temperature necessary to effect the proper result varies with the proportion of the ingredients, and ranges from 250° to something over 300°, this also being more or less modified by the time employed. - When India rubber is woven into fabrics, it is prepared for the purpose by slicing it into threads, with knives worked by machinery and kept wet. These threads are wound upon cylinders in a state of tension, and are woven into the fabric in this condition. In the early manufacture of fabrics of this kind a process technically called "shirring" was employed. The elastic threads, in a state of tension, were passed between rollers, and then between two other rollers over each of which was passed a strip of cloth, cotton, or silk. This brought the threads between the two layers of cloth, and the latter having been prepared with a coating of India-rubber cement, they were held there. One of these shirring machines, together with a machine for cutting the threads, was the invention of James Bogardus of New York, and was extensively used for a number of years.
The goods made by that process have however entirely given place to woven fabrics; and the cutters now used are single circular knives, revolving with high speed, cutting sheets wound upon cylinders, which are given a slow rotary as well as a side motion, by which the thread is cut in a spiral. - Vulcanized caoutchouc, under the name of hard rubber and ebonite, has of late years been applied to a great variety of uses. The best ebonite is made of pure caoutchouc and sulphur, subjected to a longer continued and higher degree of heat than the ordinary pliable article for springs, tubing, hose, etc. Optical and surgical instruments are composed in part and sometimes entirely of this substance, which is susceptible of taking a high degree of finish and is very strong and elastic. For the backs of brushes it has been found to be well adapted, when mixed with various mineral ingredients by which its plasticity is so affected that the brush may be made by one operation in a die, the material enclosing the tufts of bristles and holding them securely without any further fastening.
It has also been applied to the making of watch cases, buttons, combs, ornamental articles of dress, and many other uses. - The following table exhibits the condition of the manufactures of caoutchouc in the United States in the years 1860 and 1870:
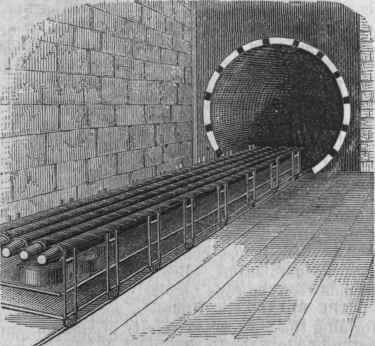
Fig. 6. - Vulcanizing Cylinder.
STATES. | ESTABLISHMENTS. | HANDS EMPLOYED. | CAPITAL INVESTED. | VALUE OF PRODUCTS. | ||||
1860. | 1870. | 1860. | 18T0. | 1860. | 1870. | 1860. | 1870. | |
Connecticut......... | 9 | 13 | 809 | 1,946 | $1,265,000 | $2,345,000 | $2,276,000 | $4,239,329 |
Maine........... | ... | 1 | ... | 9 | .... | 4,000 | ... | 31.500 |
Massachusetts......... | 5 | 16 | 298 | 1,405 | 563,000 | 1,920,000 | 803,000 | 3,183.218 |
Missouri.......... | ... | 1 | .... | 4 | ... | 2,000 | ... | 4,500 |
New Jersey......... | 5 | 12 | 817 | 807 | 870.000 | 1,034,000 | 1,303,000 | 2.224,839 |
New York........... | 7 | 10 | 757 | 1,008 | 775,000 | 1,777,000 | 1,127,750 | 3,076,720 |
Pennsylvania.......... | 1 | 1 | 8 | 1 | 5,000 | 800 | 12.000 | 1,400 |
Rhode Island.......... | 2 | 2 | 113 | 845 | 156,000 | 403,000 | 246,700 | 1,804,868 |
Total in United States.. | 29 | 56 | 2,802 | 6,025 | $3,364,000 | $7,4S5,800 | $5,768,450 | $14,566,374 |
Among the products of 1870 were 1,250,000 lbs. of car springs, 906,000 lbs. of belting and hose, 552,500 dozen braces, 5,402,666 pairs of boots, and 30,000 coats. There were consumed 8,413,320 lbs. of caoutchouc, 2,934,575 yards of cloth, 2,391,451 lbs. of cotton, and 2,900 lbs. of silk; the value of all the materials was $7,434,742. The amount of caoutchouc received at the port of New York from various countries in 1870 was about 8,500,000 lbs., and in 1871 about 9,000,000 lbs. In 1872 the imports were as follows:
From Para.......... | 5,182,751 lbs. |
" Central America.............. | 5,694,619 " |
" Mexico............... | 139,950 " |
" Cartagena........ | 267,888 " |
" India......................... | 379,200 " |
Miscellaneous....................... | 197,881 " |
Total imports........... | 11,860,929 " |
The imports of caoutchouc into Great Britain for the nine months ending Sept. 30, 1872, were 13,464,752 lbs., valued at $6,813,300.
Continue to:


