Manometer
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Manometer
Manometer (Gr.![]() , rare, and
, rare, and![]() ,measure - measurer of rarity), an instrument employed to measure the pressure exerted by a confined portion of gas or vapor. The force is usually expressed in units of atmospheric pressure, called atmospheres, which are equal to 30 inches height of a column of mercury, or nearly 15 lbs. to the square inch. It will therefore be easily seen that mechanical ingenuity may devise several forms of the instrument. These various forms may be classified under three different general forms, which act upon different principles: 1, open-air manometer; 2, confined-air manometer; 3, metallic-spring manometer. An open-air manometer is shown in fig. 1. It consists of a vessel containing mercury in which a vertical tube 5 dips. The vessel also admits a tube, a, which connects with the boiler or chamber of compressed gas or steam. Calling Boyle's or Mariotte's law correct for all pressures, if the compressed gas has a density twice as great as it would have at the ordinary atmospheric pressure, it will raise the column of mercury in the tube b 30 inches; if five times as dense, the height of the mercurial column will be 150 inches, corresponding to 75 lbs. to the square inch.
,measure - measurer of rarity), an instrument employed to measure the pressure exerted by a confined portion of gas or vapor. The force is usually expressed in units of atmospheric pressure, called atmospheres, which are equal to 30 inches height of a column of mercury, or nearly 15 lbs. to the square inch. It will therefore be easily seen that mechanical ingenuity may devise several forms of the instrument. These various forms may be classified under three different general forms, which act upon different principles: 1, open-air manometer; 2, confined-air manometer; 3, metallic-spring manometer. An open-air manometer is shown in fig. 1. It consists of a vessel containing mercury in which a vertical tube 5 dips. The vessel also admits a tube, a, which connects with the boiler or chamber of compressed gas or steam. Calling Boyle's or Mariotte's law correct for all pressures, if the compressed gas has a density twice as great as it would have at the ordinary atmospheric pressure, it will raise the column of mercury in the tube b 30 inches; if five times as dense, the height of the mercurial column will be 150 inches, corresponding to 75 lbs. to the square inch.
There may be many forms of open-air manometers, and the modifications are generally for the purpose of increasing the convenience of the apparatus by shortening the distance of the rise of the mercurial column. The multiple-branch manometer, fig. 2, is a convenient form. An iron tube is bent upon itself, forming several U-shaped flexures, terminating in a vertical tube of glass, C D, furnished with a graduated scale, and open at the top. Mercury occupies the lower flexures and portions of the tube. When the compressed steam or gas is admitted, it presses upon the mercury in the first branch, A, forcing it down, and therefore up in the second branch. If it forces A down 10 inches, the difference of level in the two branches will be 20 inches. If there are 10 single or 5 double columns, the combined height of mercury supported in column will be 100 inches, or about 6-66 atmospheres. The compressed-air manometer, fig. 3, is constructed upon the assumption that the confined air in the gauge expands and is condensed in accordance with Boyle's law. A is a U-shaped glass tube, one end of which communicates with the steam chamber, while the other end is closed. It has its flexure stopped with mercury, and a scale is attached, which is graduated by connecting the apparatus with an open-air manometer.
It will be seen that as the mercury in A rises, the pressure is doubled for every reduction of the confined air to one half its volume, so that as the column approaches the top the graduated spaces must be nearer together. The metallic-spring manometer may be constructed by having a piston press against a spiral spring, which is also connected with an index; or a flat copper tube (elliptic section), bent in a spiral, may be connected at one end with the steam chamber, and at the other with an index, as in Burdon's pressure gauge, shown in fig. 4. Increased pressure causes the spiral to uncoil, by which the index is moved over the graduated arc.
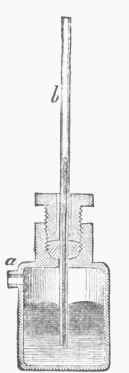
Fig. 1. - Mercurial Manometer.
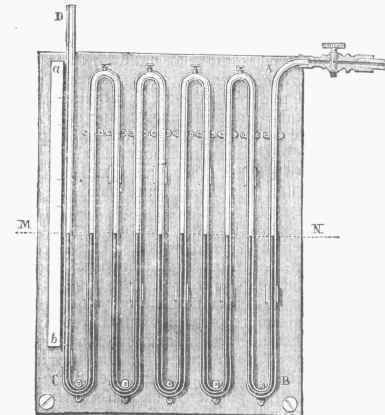
Fig. 2. - Multiple-Branch Manometer.

Fig. 3. - Compressed-air Manometer.
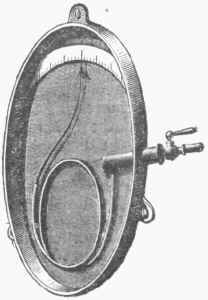
Fig. 4. - Bourdon's Pressure Gauge.
Continue to:


