The Structure Of The Developed Image. Continued
Description
This section is from the book "The Fundamentals of Photography", by C. E. K. Mees,. Also available from Amazon: The Fundamentals Of Photography.
The Structure Of The Developed Image. Continued
The swelling of a gelatine film is influenced by the temperature of the solution in which it is placed and also by the presence of other substances in the solution. A small amount of either acid or alkali will produce a considerable increase in the swelling, and since the developer is alkaline and the fixing bath is acid, both these solutions have a great tendency to swell the gelatine, especially when they are warm. On the other hand, sulphites tend to prevent swelling, so that an increase in the concentration of the sulphite in a developer or fixing bath will diminish it. An even greater aid in preventing swelling is the hardener in the fixing bath. The hardening agents used in fixing baths are the alums, which not only prevent the swelling of the gelatine temporarily but which permanently harden the structure of the gelatine so that it will not easily swell. The alum is introduced into the fixing bath so that after fixing the film will not become soft and disintegrate in washing.


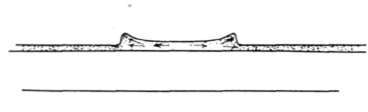

Fig. 41. The Way a Waterspot Dries.
Reticulation is due to local strains in the gelatine, and a sudden change in the temperature of solutions will sometimes produce this effect. If a film is transferred for instance from a cold fixing bath containing a hardener to very warm wash water, the whole film will sometimes pucker into tiny reticulations, a good example of which is shown in Fig. 39. If one part of the film contains much more moisture than another, the silver image itself is liable to become distorted by the movement of the gelatine, and of the silver grains in it. If a drop of water, for instance, falls on a film and this is dried rapidly, it will often produce a curious ring-shaped mark, the middle of the drop being lighter and the edge of the drop darker than the surrounding negative, Fig. 40. The explanation of this is shown in Fig. 41. The gelatine swells up where the spot of water fell on it, and as it dries again a strain is produced by the collapse of the center of the swollen spot, and so the gelatine and silver grains are pulled in to the edges of the spot and there produce the dark ring.

Appearance Of Developed Emulsion When Magnified
20 diameters
400 diameters


100 diameters
900 diameters

Fig. 42. Appearance of Emulsion After Development When Magnified.
The developed image consists of grains of silver, each grain under sufficient magnification looking like a little mass of coke, replacing one of the silver bromide crystals which were originally formed in the emulsion and keeping the same position. See Fig. 37. When we look at a negative it appears perfectly smooth to the eye, but under a small degree of magnification it begins to show an appearance of graininess.
It must not be thought, however, that with a magnifying glass we can see the silver grains themselves. The silver grains are so small that to make them visible requires powerful magnification. What we see through the magnifying glass are clumps of grains.
Suppose that an aviator is flying over country dotted with occasional woods and clumps of bushes. If he is flying near to the ground, he will be able to distinguish the separate trees and bushes. If he goes higher, he will no longer be able to see them separately but he will see them in little clumps of two and three where they are close together with the spaces where they are farther apart showing between them, and then as he goes higher still, he will no longer be able to see these small clumps, but will be able to see only the large masses of woodland or forest. In the same way when we look at a negative under a low magnification, we see the larger masses of clumps of grains, and then as we increase the magnification we see the smaller clumps of grains, and then finally at a very high magnification we see the grains themselves, Fig. 42.
These clumps of grains which we can see under low magnification are made up of grains which are not all in the same layer. This can be seen by first of all photographing an image from above and then cutting a section down through it so as to see how the grains lie one below the other. In Fig. 43 A it will be seen that the image is as much as six grains deep so that many of the clumps of grains seen in Fig. 43B are not made up of grains in the same layer but of grains in different layers, some on the top and some below.

A
Vertical Section Showing Grain Deposit.
B
Horizontal Plan of Same Grain Deposit.
Fig. 43.

Exposed 1 Unit of Time

Exposed 16 Units of Time.

Exposed 4 Units of Time.

Exposed 64 Units of Time.
Fig. 44.
The distribution of the grains in the depth of the film is interesting. It might be thought that with short exposures the image would be on the top of the film and that as the exposure was continued, the light would penetrate farther and farther into the film, making the grains in the lower layers more and more developable. This sometimes seems to be the case, but with some emulsions it is not so, as is proved by the photographs of sections shown in Fig. 44, which are cut from an N. C. film. These are fully developed so that the effect of development is eliminated, and they show that the grains are exposed at all parts of the film to an almost equal extent, though in the second and third prints there is a slight tendency for the image to be more on the top of the film. It looks as though the emulsion contains grains of various degrees of sensitiveness and the more sensitive grains are made developable first. Further, since there is certainly more light at the surface of the film, it must be a fact that the more sensitive grains are found in the lower parts of the film.

1.

2.

3.

4.
Fig. 45.
Showing Progress of Development from Surface to Base of Emulsion.

Strong or Concentrated Developer.
Weak or Diluted Developer. Fig. 46.
During development, however, there is an appreciable effect due to the penetration of the developer into the film. This is shown in Fig. 45, where it is seen that at the beginning of development only the surface of the emulsion is developed, and then as development continues the developer penetrates into the film and develops more and more deeply in it. In the case of a strong developer this effect is accentuated, because a strong developer will develop the surface to good density before it has penetrated through the emulsion, while a weak developer will penetrate at the same rate as the strong developer and will not develop so rapidly, so that with a strong developer there is a tendency for the image to be confined to the surface of the emulsion, and with a weaker developer for it to penetrate through the whole emulsion. This effect is well shown in Fig. 46, where two photographs are shown of the edge of an exposed image, the image being shown as the dark part on the left, while on the right we have the light deposit of grains due to fog. The broad black line at the bottom of each illustration represents the film on which the emulsion is coated. In the upper picture, the image was developed with a very strong developer, while in the lower picture it was developed with a much weaker developer, and it will be noted that the weak developer has penetrated right through the image to the back, while with the strong developer the image has not developed through to the back of the film, although care was taken to develop the images to the same apparent density.
There is a curious effect shown in these photographs at the point marked A, where it is seen that at the edge of the developed image the fog grains are not developed in the lower part of the film; it is as if they had been eaten away. There is no doubt that the reason for this is that the bromide liberated during development of the heavy image has prevented the fog grains close to the edge of the image from developing. In extreme cases this will sometimes surround a dense image with a white line.
Continue to:


