Zinc Chloride
Description
This section is from the book "Soldering For Workshop, Farm And Home", by John Bonert. Also available from Amazon: Soldering For Workshop, Farm And Home - Information On Soft And Hard Soldering - Projects For The Workshop Explained And Illustrated.
Zinc Chloride
Zinc chloride is the most important flux as it can be used for so many metals. It is easy to prepare and can be kept for a long time in a covered container. When zinc chloride is prepared the container must be of an acid proof material such as lead, glass or earthenware. Don't use a bottle with the top broken away. This leaves jagged points and sharp edges. The writer has seen such containers many times in tinsmith shops. It is a sure accident hazard. It is best to use two jars, the first one being used for dissolving the zinc. The clear liquid is poured into the second jar which is to be used as the permanent container. The sediment which remains in the first jar will have to be disposed of. A large discarded cold cream jar or a glass jar with a large screw top is suitable as a container. The cover of the jar if made of metal should be covered on the inside with vaseline or automobile grease to prevent corrosion.
Preparing Zinc Chloride
Enough Hydrochloric (muriatic) acid is poured into the first jar. Use only enough so that when it is poured off it will fill the jar which is to be used as the permanent container about one third full. A few small pieces of sheet zinc are cut and dropped into the acid. The acid will begin to dissolve the zinc as soon as it is dropped in. Too much zinc must not be dropped in at one time as a boiling action takes place and the acid may boil over. The zinc is added until no more will be dissolved by the acid. After the mixture has cooled and settled pour off the clear liquid into the second jar and to this add an equal amount of water. The zinc chloride is now ready to be used. Care must be taken not to inhale any of the fumes that are generated when the zinc is dissolving. The fumes are highly inflammable. The acid is also very destructive if it comes in contact with one's skin or clothes. If there is no zinc at hand the metal casing of an old discarded dry cell battery can be used as this metal is zinc.
Brush For Zinc Chloride
A good brush for applying zinc chloride can be made with a piece of copper tubing having either a one quarter or three-eighth-inch hole and about five or six inches long. If there is no copper tubing at hand a piece of sheet copper or brass may be used. It will have to be rolled or folded to make a tube that will have about a three-eighth-inch hole. Cut enough bristles from an old brush to fill the hole in the tube. The bristles should be cut about one inch long and should extend about one-half inch out of the tube. Place the tube in the vise so that about one-half inch of the tube will be flattened against the bristles when the vise is screwed up. This will keep the bristles from coming out. A brush made in this manner will last a long time if it is not allowed to remain in the zinc chloride when not in use. If sheet copper or brass is used it should be rolled or bent so the edges overlap as illustrated in Figure 1.
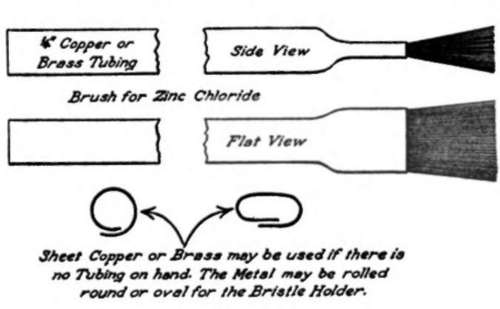
Figure 1.
Continue to:


