The Various Modes Of Transmitting Power To A Distance. Part 2
Description
This section is from "Scientific American Supplement Volumes 275, 286, 288, 299, 303, 312, 315, 324, 344 and 358". Also available from Amazon: Scientific American Reference Book.
The Various Modes Of Transmitting Power To A Distance. Part 2
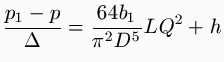 . The following are the details of the experiments: First series of experiments: Conduit consisting of cast or wrought iron pipes, joined by means of flanges, bolts, and gutta percha rings. D = 0.20 m. (8 in.); L = 4,600 m. (15,100 ft,); h= 26.77 m. (87 ft. 10 in.). 1st experiment: Q = 0.1860 cubic meter (6.57 cubic feet), at a pressure of ½(p + p), and a temperature of 22° Cent. (72° Fahr.); p = 5.60 atm., p =5.24 atm. Hence p - p = 0.36 atm.= 0.36 x 10,334 kilogrammes per square meter (2.116 lb. per square foot), whence we obtain b=0.0001697. D'Aubuisson's formula would have given p - p = 0.626 atm.; and M. Arson's would have given p - p = 0.9316 atm. 2d experiment: Q = 0.1566 cubic meter (5.53 cubic feet), at a pressure of ½(p + p), and a temperature of 22° Cent. (72° Fahr.); p = 4.35 atm., p = 4.13 atm. Hence p - p = 0.22 atm. = 0.22 X 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.0001816. D'Aubuisson's formula would have given p - p = 0.347 atm; and M. Arson's would have given p - p = 0.5382 atm. 3d experiment: Q = 0.1495 cubic meter (5.28 cubic feet) at a pressure of ½(p + p) and a temperature 22° Cent. (72º Fahr.); p = 3.84 atm., p = 3.65 atm. Hence p - p = 0.19 atm. = 0.19 X 10,334 kilogrammes per square meter (2.116 lb. per square foot); whence we obtain B = 0.0001966. D'Aubuisson's formula would have given p - p = 0.284 atm., and M. Arson's would have given p - p = 0.4329 atm. Second series of experiments: Conduit composed of wrought-iron pipes, with joints as in the first experiments. D = 0.15 meter (6 in.), L - 0.522 meters (1,712 ft.), h = 3.04 meters (10 ft.) 1st experiments: Q = 0.2005 cubic meter (7.08 cubic feet), at a pressure of ½(p + p), and a temperature of 26.5° Cent. (80° Fahr.); p = 5.24 atm., p = 5.00 atm. Hence p - p = 0.24 atm. =0.24 x 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.3002275. 2nd experiment: Q = 0.1586 cubic meter (5.6 cubic feet), at a pressure of ½(p + p), and a temperature of 26.5° Cent. (80° Fahr.); p = 3.650 atm., p = 3.545 atm. Hence p - p = 0.105 atm. = 0.105 x 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.0002255. It is clear that these experiments give very small values for the coefficient. The divergence from the results which D'Aubuisson's formula would give is due to the fact that his formula was determined with very small pipes. It is probable that the coefficients corresponding to diameters of 0.15 meter (6 in.) and 0.20 meter (8 in.) for a substance as smooth as tin, would be still smaller respectively than the figures obtained above.
. The following are the details of the experiments: First series of experiments: Conduit consisting of cast or wrought iron pipes, joined by means of flanges, bolts, and gutta percha rings. D = 0.20 m. (8 in.); L = 4,600 m. (15,100 ft,); h= 26.77 m. (87 ft. 10 in.). 1st experiment: Q = 0.1860 cubic meter (6.57 cubic feet), at a pressure of ½(p + p), and a temperature of 22° Cent. (72° Fahr.); p = 5.60 atm., p =5.24 atm. Hence p - p = 0.36 atm.= 0.36 x 10,334 kilogrammes per square meter (2.116 lb. per square foot), whence we obtain b=0.0001697. D'Aubuisson's formula would have given p - p = 0.626 atm.; and M. Arson's would have given p - p = 0.9316 atm. 2d experiment: Q = 0.1566 cubic meter (5.53 cubic feet), at a pressure of ½(p + p), and a temperature of 22° Cent. (72° Fahr.); p = 4.35 atm., p = 4.13 atm. Hence p - p = 0.22 atm. = 0.22 X 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.0001816. D'Aubuisson's formula would have given p - p = 0.347 atm; and M. Arson's would have given p - p = 0.5382 atm. 3d experiment: Q = 0.1495 cubic meter (5.28 cubic feet) at a pressure of ½(p + p) and a temperature 22° Cent. (72º Fahr.); p = 3.84 atm., p = 3.65 atm. Hence p - p = 0.19 atm. = 0.19 X 10,334 kilogrammes per square meter (2.116 lb. per square foot); whence we obtain B = 0.0001966. D'Aubuisson's formula would have given p - p = 0.284 atm., and M. Arson's would have given p - p = 0.4329 atm. Second series of experiments: Conduit composed of wrought-iron pipes, with joints as in the first experiments. D = 0.15 meter (6 in.), L - 0.522 meters (1,712 ft.), h = 3.04 meters (10 ft.) 1st experiments: Q = 0.2005 cubic meter (7.08 cubic feet), at a pressure of ½(p + p), and a temperature of 26.5° Cent. (80° Fahr.); p = 5.24 atm., p = 5.00 atm. Hence p - p = 0.24 atm. =0.24 x 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.3002275. 2nd experiment: Q = 0.1586 cubic meter (5.6 cubic feet), at a pressure of ½(p + p), and a temperature of 26.5° Cent. (80° Fahr.); p = 3.650 atm., p = 3.545 atm. Hence p - p = 0.105 atm. = 0.105 x 10,334 kilogrammes per square meter (2,116 lb. per square foot); whence we obtain b = 0.0002255. It is clear that these experiments give very small values for the coefficient. The divergence from the results which D'Aubuisson's formula would give is due to the fact that his formula was determined with very small pipes. It is probable that the coefficients corresponding to diameters of 0.15 meter (6 in.) and 0.20 meter (8 in.) for a substance as smooth as tin, would be still smaller respectively than the figures obtained above.The divergence from the results obtained by M. Arson's formula does not arise from a difference in size, as this is taken into account. The author considers that it may be attributed to the fact that the pipes for the St. Gothard Tunnel were cast with much greater care than ordinary pipes, which rendered their surface smoother, and also to the fact that flanged joints produce much less irregularity in the internal surface than the ordinary spigot and faucet joints.
Lastly, the difference in the methods of observation and the errors which belong to them, must be taken into account. M. Stockalper, who experimented on great pressures, used metallic gauges, which are instruments on whose sensibility and correctness complete reliance cannot be placed; and moreover the standard manometer with which they were compared was one of the same kind. The author is not of opinion that the divergence is owing to the fact that M. Stockalper made his observations on an air conduit, where the pressure was much higher than in gas pipes. Indeed, it may be assumed that gases and liquids act in the same manner; and, as will be [1] explained later on, there is reason to believe that with the latter a rise of pressure increases the losses of pressure instead of diminishing them.
[Transcribers note 1: corrected from 'as will we explained']
All the pipes for supplying compressed air in tunnels and in headings of mines are left uncovered, and have flanged joints; which are advantages not merely as regards prevention of leakage, but also for facility of laying and of inspection. If a compressed air pipe had to be buried in the ground the flanged joint would lose a part of its advantages; but, nevertheless, the author considers that it would still be preferable to the ordinary joint.
It only remains to refer to the motors fed with the compressed air. This subject is still in its infancy from a practical point of view. In proportion as the air becomes hot by compression, so it cools by expansion, if the vessel containing it is impermeable to heat. Under these conditions it gives out in expanding a power appreciably less than if it retained its original temperature; besides which the fall of temperature may impede the working of the machine by freezing the vapor of water contained in the air.
If it is desired to utilize to the utmost the force stored up in the compressed air it is necessary to endeavor to supply heat to the air during expansion so as to keep its temperature constant. It would be possible to attain this object by the same means which prevent heating from compression, namely, by the circulation and injection of water. It would perhaps be necessary to employ a little larger quantity of water for injection, as the water, instead of acting by virtue both of its heat of vaporization and of its specific heat, can in this case act only by virtue of the latter. These methods might be employed without difficulty for air machines of some size. It would be more difficult to apply them to small household machines, in which simplicity is an essential element; and we must rest satisfied with imperfect methods, such as proximity to a stove, or the immersion of the cylinder in a tank of water. Consequently loss of power by cooling and by incomplete expansion cannot be avoided. The only way to diminish the relative amount of this loss is to employ compressed air at a pressure not exceeding three or four atmospheres.
Continue to:


