An Electrical Centrifugal Machine For Laboratories
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
An Electrical Centrifugal Machine For Laboratories
By ALEXANDER WATT, F.I.C., F.C.S.
The late Dr. Mohr7 of Bonn, advocated the use of a centrifugal machine as a means of rapidly drying crystals and crystalline precipitates; but although they are admirably adapted for that purpose, centrifugal machines are seldom seen in our chemical laboratories.
The neglect of this valuable addition to our laboratory apparatus is probably owing to the inconvenience involved in driving the machine at a high speed by means of the ordinary hand driving gear, especially when the rotation has to be maintained for a considerable length of time. It occurred to me, therefore, that by attaching the drum or basket of the machine (or the rotating table of Mohr's apparatus) directly to the spindle of an electro-motor, the difficulty of driving might be got over, and at the same time a combination of great efficiency would result, as the electro-motor, like the centrifugal machine, is most efficient when run at a high speed. The apparatus shown in the sketch consists essentially of a perforated basket, A, which is slipped on to a cone attached to the spindle, S, of an electro-motor, and held in position by the nut, D. The casing, B, with its removable cover, C, serves to receive the liquid driven out of the substance being dried. A flat form of the ordinary Siemens H armature, E, revolves between the poles, P, of the electro-magnets, M, which are connected by means of the base plate, I. The brass cross-bar, G, carries the top bearing of the spindle, S, and prevents the magnet poles from being drawn together.
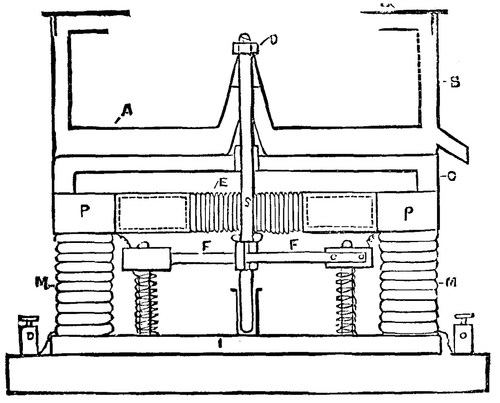
From four to six cells of a bichromate battery or Faure secondary battery furnish sufficient power to run the machine at a high speed. An apparatus with a copper basket four inches in diameter has been found extremely useful in the laboratory for drying such substances as granulated sulphate of copper and sulphate of iron and ammonia, but more especially for drying sugar, which when crystallized in very small crystals cannot be readily separated from the sirupy mother-liquor by any of the usual laboratory appliances. For drying substances which act on copper the basket may be made of platinum or ebonite; in the latter case, owing to the increased size of the perforations, it may be necessary to line the basket with platinum wire gauze or perforated parchment paper.
[6]Paper read before Section B, British Association, Aberdeen meeting.[7]"Lehrb. d. Chem. Analyt. Titrirmethode," 3d ed., 1870, p. 684.
Continue to:


