Isochromatic Photography
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
Isochromatic Photography
By FRED. E. IVES.
It is well known that the ordinary photographic processes do not reproduce colors in the true proportion of their brightness. Violet and blue photograph too light; green, yellow, orange and red, too dark. For a long time it was believed to be impossible to remedy this defect; and even when it became known that bromide of silver could be made more sensitive to yellow and red by staining it with certain dyes, the subject received very little attention, because it was also known that the increase of sensitiveness was too slight to be of practical value in commercial photography.
Dr. H.W. Vogel, who was one of the first, though not the first, to devote attention to this subject, announced, in 1873, that he had succeeded in making a yellow object photograph lighter than a blue or violet one, by using a silver-bromide plate stained with coraline, and exposed through a yellow glass. The plate showed no increased sensitiveness to red, and the experiment, although of considerable scientific interest, did not indicate a practically useful process.
In the spring of 1878 I became interested in this subject, and tried to discover a method of producing plates which should be sensitive to all colors, and capable of reproducing them in the true proportion of their brightness. I commenced by trying nearly all the color sensitizers which had already been suggested, in order to learn which was the best, and then, if possible, why it was the best, as a guide to further research. Chlorophyl was the only thing I tried which was sufficiently sensitive to red to offer any encouragement in that direction; but the solution which I obtained was weak and unstable, and far from being a satisfactory color sensitizer. Hoping to obtain a better solution with which to continue my experiments, I made extracts from many kinds of leaves, and found that a solution from blue myrtle leaves looked better and kept better than any other, and when it was applied to the silver-bromide plates they became remarkably sensitive, not only to all shades of red, but also to orange, yellow, and green.
By placing in front of the lens a color-screen consisting of a small glass tank containing a weak solution of bichromate of potash, to cut off part of the blue and violet light, I obtained, with these chlorophyl plates, the first photographs in which all colors were reproduced in the true proportions of their brightness. But my chief desire at that time was to realize a method of producing from any object in colors a set of three negatives, in one of which the shadows should represent the blue of the original, in another the yellow, and in another the red, in such a manner that transparent pigment prints from these negatives - blue, yellow, and red - would, when superimposed on a white surface, represent not only the lights and shadows, but also the colors of the object. This had already been attempted by others, who failed because their plates were not sufficiently sensitive to red and yellow.
Having succeeded perfectly in my undertakings, I published my discovery in 1879,[2] explaining how to prepare and use the chlorophyl plates, in connection with the yellow screen, for the purpose of securing correct photographs of colored objects.[3]
So far as I know, nobody tried the process. Nearly five years later Dr. Vogel announced that, after eleven years of investigation, he had at last realized a successful process of this character, and that this new process of his was the "solution of a problem that had long been encompassed with difficulty." This publication attracted a great deal of attention, and gave me occasion to again call attention to my process,[4] and point out that it was not only the first practical solution of this problem, but the only truly isochromatic process ever discovered. Dr. Vogel's new process was not only no better in any respect, but the plates were insensitive to scarlet and ruby-red, and therefore would not photograph all colors in the true proportion of their brightness.
My method consists in treating ordinary collodio-bromide emulsion plates with blue myrtle chlorophyl solution, exposing them through the yellow screen, and then developing them in the usual manner. The emulsion which I have employed is made with an excess of nitrate of silver, which is afterward neutralized by the addition of chloride of cobalt; it is known as Newton's emulsion. I now prepare the chlorophyl from fresh blue myrtle leaves, by cutting them up fine, covering with pure alcohol, and heating moderately hot; the leaves are left in the solution, and some zinc powder is added, which helps to keep the chlorophyl from spoiling. I have a bottle of this solution which was prepared about six months ago, and now appears to be as good as when first made.[5] A glass plate is flowed with the emulsion, and as soon as it has set, the chlorophyl solution is applied for a few seconds, after which the plate is washed in pure water until smooth, when it is ready for exposure.
My color-screen consists of a small plate-glass tank, having a space of 3/16 of of an inch between the glass, filled with a solution of bichromate of potash about one grain strong. I place the tank in front of the lens, in contact with the lens-mount. The advantage of this tank and solution is that it can be more easily obtained than yellow plate glass, and the color can be adjusted to meet any requirement.
The plates require about three times as much exposure through the yellow screen as without it, and may be developed with the ordinary alkaline pyro-developer.
IVES' PROCESS OF ISOCHROMATIC PHOTOGRAPHY.
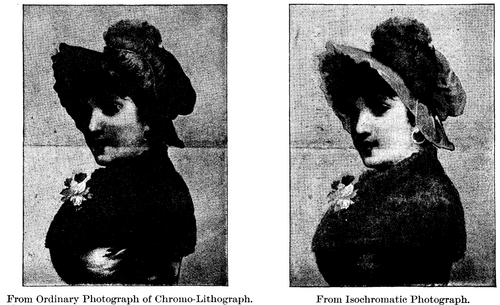
In order to illustrate the value of this process, I made two photographs of a highly-colored chromo-lithograph, representing a lady with a bright scarlet hat and purple feather, a yellow-brown cape and a dark-blue dress. One, by the ordinary process, represents the blue as lighter than the yellow-brown, the bright scarlet hat as black, and the purple feather as nearly white. The other, by the chlorophyl process, reproduces all colors in nearly the true proportion of their brightness, but with a slight exaggeration of contrast produced purposely by using a too-strong color solution in the small tank.
I also made two landscape photographs, one by the ordinary process, and the other by the chlorophyl process, exposing them simultaneously. In the ordinary photograph, distant hills are lost through overexposure, yet the foreground seems underexposed, and yellow straw-stacks and bright autumn leaves appear black. In the chlorophyl photograph, the distant hills are not overexposed, nor is the foreground underexposed; the yellow straw-stacks appear nearly white, and bright autumn leaves contrast strongly with the dark green about them.
To test the relative color-sensitiveness of plain emulsion plates, plates stained with eosine, and plates stained with the blue-myrtle chlorophyl, I exposed one of each kind through the same yellow screen, giving each five minutes exposure, on the same piece of copy, which was the chromo-lithograph already described. The plain emulsion plate showed only the high lights of the picture, after prolonged development. The eosine plate was underexposed, but brought up everything fairly well except the scarlet hat, which came up like black. The chlorophyl plate was overexposed, brought out all colors better than the eosine plate, and gave full value to the bright scarlet of the hat, the detail in which was beautifully rendered.
Dr. Vogel advanced the theory that silver-bromide is insensitive to yellow and red, because it reflects or transmits those colors; and that it becomes sensitive when stained, because of the optical properties of the dyes. He afterward admitted that only such dyes as are capable of entering into chemical combination with the silver-bromide proved capable of increasing its sensitiveness to color, but he held to the theory that the optical properties of the compound were the cause of its color-sensitiveness.
I have shown that the color-sensitiveness can be produced by treatment with an organic compound which has none of the optical properties characteristic of dyes; and that chlorophyl, which absorbs only red light, greatly increases the sensitiveness also to yellow and green. There is, therefore, good reason to doubt if the color-sensitiveness is ever due to the optical properties of the dye or combination.
Attempts have been made to produce isochromatic gelatine dry plates which, while many times more sensitive to white light than my chlorophyl plates, shall also show the same relative color-sensitiveness. Such plates would be very valuable but for one fact: it would be necessary to prepare and develop them in almost total darkness. Gelatine bromide dry plates extremely sensitive to yellow, but comparatively insensitive to red, might be used to advantage in portrait and instantaneous photography, because they could be safely prepared and developed in red light; but when truly isochromatic photographs are required, the time of exposure must be regulated to suit the degree of sensitiveness to red, which cannot safely be made greater than I have realized with my chlorophyl process.
[1]Read at the stated meeting of the Franklin Institute, March 18, 1885.[2]Philadelphia Photographer, December, 1879, p. 365.[3] I intended this publication to be a very full and explicit one, and it was sufficiently so to be perfectly understood by most who saw it; but some may think I did not sufficiently emphasize the importance of using the particular kind of chlorophyl which I mentioned. In a brief communication to the editor of the Photo. News, in 1883, I described some experiments with eosine as a color sensitizer, and then called attention to the superiority of blue-myrtle chlorophyl for this purpose, stating that I had not been able to secure such results with any other kind of chlorophyl, and that a fresh solution from fresh leaves must be used to secure the greatest possible degree of sensitiveness. See Photo. News, Nov. 1883, p. 747.[4]Photo. News, London, September 5, 1884, p. 566, and Year Book of Photography for 1885, p. 111.[5]I originally recommended chlorophyl extracted from dried leaves, because I had not yet learned how to preserve the solution for more than a few weeks; and at some seasons it would be difficult, if not impossible, to obtain fresh leaves.The tea organifier which I recommended is also a color sensitizer, and when it is used in connection with the chlorophyl from dried leaves the plates are as sensitive to red as can be safely prepared and developed in the light of an ordinary photographic "dark-room." Plates prepared with chlorophyl from fresh leaves do not require treatment with the tea organifier to secure this degree of sensitiveness. Recently I have used the tea organifier and some other sensitizers, in connection with the solution from fresh myrtle-leaves, and in this way have produced plates having such an exalted color sensitiveness as to be unmanageable in ordinary "dark-room" light. Possibly, such plates might be prepared and developed in total darkness, by the aid of suitable mechanical contrivances, but I am not sure that they would work clear even then, because they appear to be sensitive to heat as well as to light.
Continue to:


