101. Eucalyptus odorata
Description
This section is from the book "A Research On The Eucalypts Especially In Regard To Their Essential Oils", by Richard T. Baker, Henry G. Smith. Also available from Amazon: A Research On The Eucalypts And Their Essential Oils.
101. Eucalyptus odorata
(Behr. in Linnæa, XX, 657.) Box-tree or Peppermint.
Systematic. - A small tree, or "Mallee," bark "Box"-like. Abnormal leaves linear to broadly lanceolate, dull, often glaucous or bluish green. Normal leaves narrow lanceolate, rigid, of a dull green or somewhat shining; intra-marginal vein somewhat removed from the edge; venation oblique, about 30° from the mid-rib. Peduncles mostly axillary, 1/4 to 1/2 inch long, bearing about seven almost sessile flowers. Buds clavate; calyx tube tapering gradually to peduncle and about twice as long as the conical obtuse operculum.
Fruit. - Cylindrical to sometimes pyriform, almost sessile, more or less angular; rim countersunk; valves scarcely or not exserted; about 4 lines long and up to 3 lines broad.
It is very difficult at times to differentiate these fruits from those of E. dumosa or perhaps E. paniculata
Habitat. - New South Wales; Victoria; and South Australia.

REMARKS. - Behr's species is acknowledged in Bentham's "Flora Australiensis" and Mueller's "Eucalyptographia," and later by Maiden in his "Critical Revision of the Eucalypts," vol. ii, Part I, p. 26, as well as in the Trans. Roy. Soc. S.A., 1903, where he goes fully into the synonymy of the species.
With most of this latter synonymy we are not in accord, especially in placing E. Lansdowniana under E. odorata. Brown, in his " Florest Flora of South Australia," figures both species and if any reliance can be placed in delineation, greater differences could not be illustrated. Brown's figure is what we regard as E. Woollsiana.
ESSENTIAL OIL. - Material for distillation was collected at the Kingscote end of Kangaroo Island in July, 1911. This species is considered by Eucalyptus oil distillers of Kangaroo Island to be of equal value with E. cneorifolia for oil production.
The origin of the name "Peppermint" for this species could not be traced, and it seems to be altogether a misnomer, as the ketone of peppermint odour (piperitone) appears to be absent in this oil, the characteristic high-boiling constituent being aromadendral. The name "Peppermint" is also given to Eucalyptus species in New South Wales, the oils of which do not contain piperitone, nor do their oils have a peppermint odour. The oils of the true "Peppermint" Eucalyptus species usually contain the terpene phellandrene, piperitone being also a characteristic constituent.
Plate LIX
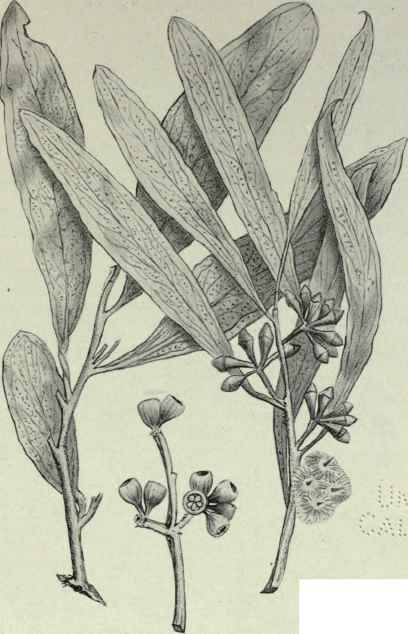
R. T. Baker, del.
Eucalyptus Odorata, Behr. Box Tree, Peppermint
The yield of oil was 1.87 per cent., but as the material was collected in the winter this may be taken as the minimum yield. The crude oil was orange-brown in tint, indicating but a trace of phenol, and had an odour similar to those belonging to the cineol-pinene group, with a secondary one indicating aroma-dendral. Volatile aldehydes were pronounced. The oil was rich in cineol, and phellandrene was absent.
The crude oil had specific gravity at 15° C. = 0.9193; rotation aD - 1.1o; refractive index at 200 = 1.4639, and was soluble in 1 1/4 volumes 70 per cent, alcohol. The lævo-rotation shown by this oil is due to the aromadendral. The saponification number for the esters and free acid was 8.5.
On rectification a few drops of acid water with some aldehydes came over below 1730 C. (corr.), showing the comparative absence of the low-boiling terpenes. Between 173-1850, 82 per cent, distilled; between 185-2150, 8 per cent, came over, and between 215-2300, 4 per cent, distilled. These fractions gave the following results: -
First fraction, sp. gr. at 150 C. | = | 0.9153; rotation aD - 0.1°; refractive index at 200 = 1.4619. | |||
Second | " | " | " | = | 0.9161; rotation aD - 0.7°; refractive index at 200 = 1.4639. |
Third | " | " | " | = | 0.9219; rotation aD - 11.7°; refractive index at 200 = 1.4704. |
The high lævo-rotation of the third fraction is due to the aromadendral. The cineol was determined by the resorcinol method in the oil distilling below 185°; when calculated for the crude oil, the result was 86 per cent. By the phosphoric acid method it was 63 per cent. (See also further analysis under "Quantitative Determination of Cineol.")
The rectified oil was slightly yellow in tint, due to the particular phenol present.
There is a strong resemblance between the oil of this Eucalypt and that of "Narrow Leaf," E. cneorifolia, and for commercial purposes no marked differences could be detected between the products of the two species.
The results obtained with the oil of this Eucalpyt were published by us in the Trans. Roy. Soc, South Australia, 1916.
Continue to:


