Australol
Description
This section is from the book "A Research On The Eucalypts Especially In Regard To Their Essential Oils", by Richard T. Baker, Henry G. Smith. Also available from Amazon: A Research On The Eucalypts And Their Essential Oils.
Australol
This crystallised phenol could be best prepared from the oils of the typical "Boxes," such as E. hemiphloia, E. Woollsiana, etc. We utilised the former of these species because it grows plentifully in the immediate neighbourhood of Sydney.
The phenolic bodies were first prepared in the manner described above, and afterwards distilled under reduced pressure. As australol boils at a considerably lower temperature than tasmanol it was not difficult to obtain the phenol fairly pure by fractionation under reduced pressure.
Australol boils at 115-1160 C. under 10 millimetres pressure, and shows a tendency to remain for a long time in the liquid condition; if, however, the minutest particle of crystal is added, the whole mass instantly solidifies.
The specific gravity at 200 C. was 0.9971; refractive index at the same temperature = 1.5195, and was optically inactive. The phenol was best purified from solution in xylene, as it crystallised well from that solvent. It remained more or less as a varnish when dissolved in alcohol, chloroform, acetone and the usual organic solvents. The long thin prism crystals (enlarged 45 diameters), as crystallised from xylene, are well shown in the accompanying photograph. They exhibit the peculiarity of rounded ends to the prisms.
The phenol sublimes splendidly, and is unaltered by this treatment; it was thus possible to obtain it quite pure. It was soluble in ammonia, but not in sodium carbonate or bicarbonate.
Australol melts at 62° C, and has a tendency to remain in the super-fused condition for some time if not seeded. It forms a benzoyl compound which melts at 72-73°, when crystallised from petroleum ether.
In alcoholic solution ferric chloride gives a fleeting greenish yellow colour at first, quickly changing to bright yellow. The phthalein fusion gives an orange-coloured solution, of little distinctive value. Concentrated sulphuric acid gives no colouration, but nitric acid forms an orange-coloured compound.
Australol has an odour reminding one of ordinary phenol, and is very caustic, destroying the skin in exactly the same manner as does phenol. It is also very volatile, and if allowed to remain in the air soon "entirely volatilises. It is an unsaturated phenol giving a liquid bromide.
Plate LXXXVII
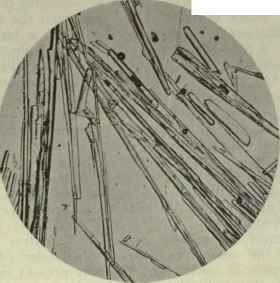
Australol.
The crystallised phenol of Eucalyptus Oils. x 45.
The formula is C9H12O, as indicated from the following analyses: -
(a) From the crystallised phenol: -
0.1589 gram. gave 0.4589 gram. CO2 and 0.1288 gram. H2O. C. = 78.7, and H = 9.0 per cent.
(b) From the sublimed phenol: -
0.1386 gram. gave 0.4004 gram. CO2 and 0.1114 gram. H2O.
C. = 78.8 and H. = 8.93 per cent.
C9.H12O requires 79.4 per cent. C. and 8.82 per cent. H.
A molecular weight determination by the cryoscopic method, using benzene as solvent, gave 142.
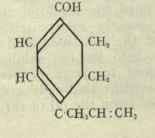
The refractive index, as previously stated, was 1.5195 at 200 C. The molecular refraction calculated from this for a C9H12O molecule with three double bonds is 41.74; found 41.50. The phenol is unsaturated and evidently one double bond is in the side chain; this, most probably, has the allyl grouping, as it formed no colour reaction with picric acid. Two double bonds thus occur in the nucleus.
Australol thus appears to be dihydro-ρ-allylphenol, and to have the following probable molecular structure; -
Continue to:


