Citronellal. Continued
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Citronellal. Continued
Another method of separation is based on the property of the citronellal to combine only with a concentrated solution of sodium sulphite and sodium bicarbonate, whereas citral also combines with dilute solutions.2)
Since methylheptenone does not combine with even a concentrated solution of sodium sulphite and sodium bicarbonate, this reagent also serves as a means to separate citronellal from any admixture of methylheptenone.3)
As with citronellol, so with citronellal, there is a difference of opinion concerning the identity of the products from different sources. Whereas e.g. Tiemann and Schmidt, also Schimmel & Co., regarded them as identical, Barbier and Bouveault1) claimed them to be isomeric. They state that citronellal from citronella oil has a different constitution (the first formula, p. 414) from that obtained upon the oxidation of citronellol from rose oil (the second formula, p. 414). To the latter they apply the name "rhodinal". They base their claims on the rearrangement of rhodinal to menthone, whereas citronellal rearranges itself to isopulegol.
1) Tiemann, Berl. Berichte 31 (1898), 3305. 2) Tiemann, Berl. Berichte 82 (1899), 815. 3) Ibidem 834.
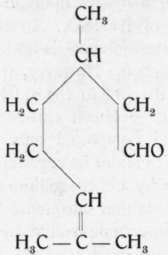
Rhodinal
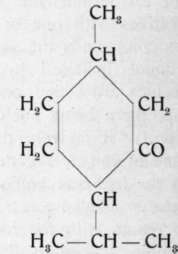
Menthone

Citronellal

Isopulegol.
The change of rhodinal to menthone is regarded as non-proven by Tiemann and Schmidt. 2) According to the examinations of these investigators, the rhodinal semicarbazone described by Barbier and Bouveault is a mixture of racemic citronellal semi-carbazone and little Isopulegone semicarbazone, whereas their supposed menthone semicarbazone is racemic Isopulegone semi-carbazone. When hydrolyzed with hydrochloric acid, Isopulegone is regenerated which has an odor resembling that of menthone. It is on this that the erroneous assumption of the French scientists is said to be based.
1) Compt. rend. 122 (1896), 737; Bouveault and Gourmand, ibidem 138 (1904), 1699; Bouveault, Bull. Soc. chim. III. 23 (1900), 458. 2) Berl. Berichte 30 (1897), 38.
The observations made by Harries and Himmelmann,1) while studying the action of ozone on citronellal, seem to indicate that the latter is not a chemical unit but a mixture of two isomeric aldehydes to which the two formulas, given at the beginning of this chapter, may be assigned. In the commercial products from different sources, the ratio of these two isomers varies. This assumption would seem to throw light on much that was previously little understood.
When carefully oxidized with silver oxide, citronellal yields the corresponding oily citronellic acid,2) C10H18O2. Energetic oxidation yields the same products obtained from citronellol, viz., acetone and /i-methyladipic acid.3) With hydroxylamine, citronellal yields a liquid oxime which when dehydrated, yields the nitrile of citronellic acid.4) The phenylhydrazone is likewise oily. So far as present observations go, the semicarbazone obtained with semicarbazide is a chemical unit and hence is well suited for the identification of citronellal. It is obtained quantitatively when an alcoholic solution of the aldehyde is shaken with a solution of semicarbazide hydrochloride and sodium acetate. When recrystallized from chloroform or ligroin it is obtained in white laminae which melt at 82,5°.5) The racemic modification melts at 96°, the thiosemicarbazone at 54 to 55°.
Like citral, so citronellal yields with pyrotartaric acid and B-naphthylamine a naphthocinchoninic acid,6) which can be utilized for purposes of identification. It is prepared like the corresponding citral derivative. The crude a-citronellyl-B-naphthocinchoninic acid is recrystallized from alcohol containing hydrogen chloride. The hydrochloride is dissolved in ammonia and the ammonium salt decomposed by acetic acid. The compound thus purified crystallizes from dilute alcohol in colorless needles that melt at 225°. When heated beyond its melting point, the acid splits off carbon dioxide and is converted into citronellyl-,B-naphtho-quinoline, a base that crystallizes from dilute alcohol or ligroin in the form of needles with a silky lustre and melts at 53°.
1) Berl. Berichte 41 (1908), 2187.
2) Semmler, Berl. Berichte 24 (1891), 208; 26 (1893), 2256.
3) Tiemann and Schmidt, Berl. Berichte 29 (1896), 908.
4) Semmler, Berl. Berichte 26 (1893), 2255.
5) Tiemann and Schmidt, Berl. Berichte 30 (1897), 34; Tiemann, ibidem 31 (1898), 3307.
6) Doebner, Berl. Berichte 27 (1894), 2025.
More rapid, however, than the identification by means of the naphthocinchoninic acid, is that by means of the semicarbazone (see above).
Upon condensation with acetone, citronellal yields citronellyl-idene acetone which yields a semicarbazide-semicarbazone that melts at 167o.1) Citronellylidene cyanacetic acid,2) prepared like the corresponding citral compound, melts at 137 to 138° and yields a characteristic, difficultly soluble sodium salt that can be utilized for the separation of citral.
Continue to:


