Coumarin
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Coumarin
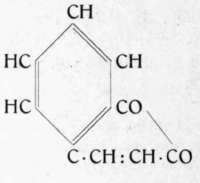
Coumarin (o-hydroxycinnamic acid anhydride), C9H602, the odoriferous principle of the "Waldmeister" (Asperula odorata), is widely distributed in the vegetable kingdom. It was first observed in the tonka beans, the fruit of Dipteryx odorata in which it occurs in largest amount. Later is was found in the "Wald-meister", also in dates, in Peru balsam, in the salikounda beans of Copeifera Salikounda, in lavender oil, in the bulbs of Vitis sessilifolia, in the resin and bark of Ceratopetalum apetalum also in the following plants: Adiantum pedatum, A. peruvianum, A. trapeziforme, Anthoxanthum odoratum, Cinna arundinacea, Hierochloa alpina, H. australis, H. borealis, Milium effusum, Aceras anthropophora, Angraecum fragrans, Nigritella angusti-folia, Orchis fusca, Orchis militaris (?), Herniaria glabra, Achlys triphylla, Prunus Mahaleb, Ruta graveolens, Dipteryx odorata,
D. oppositifolia, D. pteropus, Melilotus alba, M. altissima, M. hamata, M. leucantha, M. officinalis, Chrysophyllum imperial is, Peristrophe an gusti;folia, Alyxia stellata, Galium tri-florum, Spermacoce semierecta, Liatris odoratissima, L. spicata, Ageratum mexicanum, A. conyzoides, Eupatorium Ayapana and
E. africanum (?).1) As melilotate, coumarin also occurs in Lind-saya cultrata and in Melilotus officinalis. Apparently coumarin does not always exist as such in the plant, but is frequently formed by ferment action since its odor often becomes noticeable only after drying. Synthetically it was obtained by Perkin2) by the action of acetic acid anhydride and sodium acetate on salicylic aldehyde. However, it is also obtained by other methods and in recent years has constituted an important article of chemical industry.
Coumarin forms colorless, shiny laminae or rhombic prisms and possesses a pleasant, spicy odor and a bitter taste. In extreme dilution its odor resembles that of new mown hay. According to Zwenger and Dronke it melts at 670.1) Schimmel & Co. observed the melting point of 69 to 70° in connection with the best commercial products. It sublimes without decomposition and boils at 290,5 to 291 °.2)
1) Literature: Bley, Arch, der Pharm. 142(1857), 32; Poulsen, Bot.Zentralbl. 15 (1883), 415; Lojander, Just Jahresbericht 1887, Bd. I. 181; Molisch and Zeisel, Berichte d. deutsch. botan. Gesellschaft 6 (1888), 353; Greshoff, Berichte d. deutsch. pharm. Ges. 9 (1899), 214; Peckolt, Zeitschr. d. allg. osterr. Apoth. Ver. 31 (1893), 829; Molisch, Apotheker Ztg. 17 (1902), 45, 137; Busse, Berichte d. deutsch. pharm. Ges. 14 (1904), 205; Peckolt, Ibidem 45; Senft, Pharm. Zentralh. 45 (1904), 599; Tschirch, Ibidem 46 (1905), 803; Schimmel's Bericht April 1889, 46; October 1889, 58; Report of Schimmel & Co. April 1890, 66; October 1900, 79; April 1902, 93.
2) Berl. Berichte 8 (1875), 1599.
Coumarin dissolves difficultly in cold, somewhat more readily in hot water, easily in alcohol, and ether and in volatile and fatty oils. As to its solubility in alcohol of different strengths, Schimmel & Co. have made the observations recorded in the following table.8)
100 parts alcohol | at 0° C. | atl6tol7°C. | at 29 to 30° C. |
of 90 vol.-percent. | 7,1 parts | 13,7 parts | 42,5 parts |
of 80 vol.-percent. | 6,0 parts | 12,3 parts | 38,3 parts |
of 70 vol.-percent. | 4,4 parts | 9,1 parts | 26,0 parts |
of 60 vol.-percent. | 3,2 parts | 6,0 parts | 16,0 parts |
of 50 vol.-percent. | 1,7 parts | 3,4 parts | 8,9 parts |
of 40 vol.-percent. | 0,7 parts | 1,5 parts | 3,9 parts |
of 30 vol.-percent. | 0,3 parts | 0,6 parts | 1,7 parts |
of 20 vol.-percent. | 0,2 parts | 0,4 parts | 0,8 parts |
of 10 vol.-percent. | 0,15 parts | 0,25 parts | 0,5 parts |
100 parts water | 0,12 parts | 0,18 parts | 0,27 parts |
When boiled with concentrated potassa solution, coumarin is hydrolized to o-coumaric acid, HOC6H4CH :CHCOOH, which melts at 207 to 208°. When fused with potassium hydroxide it yields salicylic acid (m. p. 156 to 157°). When reduced with sodium amalgam, melilotic acid, HO • C6 H4 CH2 • CH2 - COOH (m. p. 81°) is obtained, whereas reduction with sodium and alcohol yields a phenol alcohol, o-hydroxydihydrocinnamic alcohol, the benzoyl compound of which melts at 99 to 1000.1) Bromine adds to form dibromcoumarin melting at 100°.2)
1) Liebig's Annalen 123 (1862), 148.
2) Perkin, Liebig's Annalen 147 (1868), 232.
3) Report of Schimmel & Co. April 1899, 66.
Coumarin and its solutions should be kept in a cool, dark place. In the course of even a few weeks light colors it yellow with polymerization to hydrocoumarin melting at 262°,3) which is an odorless compound difficultly soluble in alcohol.
When ascertaining its purity, the determination of its melting point is of first importance, then that of its solubulity in various solvents. Adulteration with acetanilid has been observed repeatedly. A method for the quantitative determination of coumarin in the presence of vanillin and acetanilid has been worked out by Winton and Bailey.4)
Continue to:


