Furfurol
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Furfurol
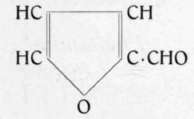
Its presence in volatile oils is possibly due to the united action of heat and acid on the cell-membrane and other carbo-hydrates of the crude materials subjected to distillation in the presence of water.1)
In as much as this aldehyde is relatively soluble in water (in 11 parts at 13°) it is contained mostly in the aqueous distillate or in the first fraction of the oil.
Furfurol has been found in the following oils, viz., pine tar oil, oil of cade, the first fraction of orris oil, clove oil,2) oil of Ceylon cinnamon, petitgrain oil, in the oil of Apocynum androsadmifolium, in the aqueous distillates from oils of cypress, savin, vetiver, orris, West Indian sandalwood, ambrette seeds, cloves, clove stems, caraway, and in the first distillate of bay and lavender oils. As a matter of interest, it may here be mentioned that E. Erdmann3) observed furfurol and furfuric alcohol in the volatile oil of the roasted coffee beans.
The boiling point of the pure compound is 160,5° (742 mm.); its density d 20o/4o 1,1594. When oxidized it yields pyromucic acid, m. p. 132 to 133°. Of its derivatives the following may here be mentioned, viz., the phenyl hydrazone, m. p. 97 to 98°, the semicarbazone, m. p. 197°, and the semioxamazone, m. p. 264°. It can be identified most conveniently by means of the well-known color reaction with p-toluidine or auiline hydrochloride.
1) Report of Schimmel & Co. October 1899, 36.
2) Oil of cloves also contains a-methyl- and a dimethyl furfurol.
3) Berl. Berichte 35 (1902), 1851.
Continue to:


