Changes The Blood Undergoes In The Lungs
Description
This section is from the book "A Manual Of Physiology", by Gerald F. Yeo. Also available from Amazon: Manual Of Physiology.
Changes The Blood Undergoes In The Lungs
In order to understand how the oxygen and the carbonic acid pass to and from the blood in the pulmonary capillaries we must know the relationship of these gases to the blood in the arterial and venous sides of the circulation.
In the chapter on the blood (pp. 243, 244) it is stated that both the oxygen and the carbon dioxide can be removed from the blood by the mercurial air pump, and that the greater part of these gases are chemically united with some of the constituents of the blood, and that a different quantity of each gas is found in arterial and venous blood. Now that we know the change from the venous to the arterial condition to take place during the passage of the blood through the pulmonary capillaries, where it is exposed to the air, we may assume that the acquisition of oxygen and the loss of C02 form the essential difference between venous and arterial blood.
From either kind of blood about 60 volumes of gas may be extracted from every 100 volumes of blood with the mercurial gas pump. The composition of this gas varies considerably in venous, but not very much in arterial blood. An average is given in the following table: -
0 vols. %. | CO vols. %. | N vols. %. | |
Arterial,...... | 20 | 39 | 1-2 |
Venous (about), . . | . . 8-10 | 46-50 | 1-2 |
The more rapidly, after bleeding, the gases are removed, the greater is the proportion of O that can be obtained, as delay allows some of it to combine with easily oxidized substances in the blood itself. The amount of oxygen varies in different parts of the venous system. In the blood of an animal dying of slow asphyxia only traces of oxygen can be found, and these soon disappear after death.
The proofs that O is, for the most part, in chemical combination with the haemoglobin of the red blood corpuscles, and not merely absorbed, as one might be led to suppose from its coming away when the pressure is reduced, are numerous and satisfactory.
1. When arterial blood is submitted to gradual diminution of pressure in the mercurial air pump the oxygen does not come away in accordance with the established law of the absorption of gases (Henry-Dalton) by coming off in proportion to the diminution of the pressure. At first only traces appear (probably the small amount really dissolved), and when the pressure has been reduced to a certain point, about one-fifth of that of the atmosphere, the oxygen comes off suddenly; after which little more can be obtained by further reduction of pressure. Haemoglobin combines with O in the same way, very rapidly at first, even when the pressure is low.
2. If the oxygen were only in a state of absorption, the blood, while passing through the pulmonary capillaries, could only take up about 0.4 volume per cent., which would be inadequate for life. We know that the quantity of O going to the blood from the air in the alveoli cannot well be explained on physical grounds alone; and when an animal dies of asphyxia from want of ventilation in a limited space, all the O of the air in the space is absorbed. Since the partial pressure of the O in the chamber falls to zero while some still exists in the haemoglobin, it cannot be the pressure which makes the O pass into the blood.
3. Another conclusive proof that the union of the O with the haemoglobin is really a chemical one, is given by the spectroscopic examination of a haemoglobin solution. When deprived of its O, and after the admixture of the air, quite dissimilar spectra are seen, as already pointed out in Chapter xiv (Blood Corpuscles). (Fig. 155, p. 3570)
4. The amount of O taken up by the blood is not always in proportion to the pressure of that gas, but rather to the amount of haemoglobin in the blood; and we therefore find the adequacy of the respiratory function of the blood going hand in hand with its richness in haemoglobin, and thus the "shortness of breath" of anaemic and chlorotic individuals is explained.
5. The oxygen can be displaced by the chemical union of other gases with the haemoglobin.
Our knowledge concerning the relation of the C02 to the constituents of the blood is less definite.
It does not all exist as a mere physical solution, for it comes off irregularly under the air pump, and does not exactly obey the Henry-Dalton law of the absorption of gases. Part comes off easily and part with difficulty. It is not associated with the corpuscles, for more of this gas can be obtained from serum than from a like quantity of blood. It is more easily removed from the blood than from the serum, a certain proportion (about 7 per cent, of the whole) remaining, in the serum in vacuo, until dissociated by the addition of an acid or a piece of clot containing corpuscles. If bicarbonate of soda be added to blood from which all the gas has been removed, still more C02 can be pumped out, from which it would appear that something exists in the blood capable of dissociating C02 from sodium bicarbonate.
It has been suggested that the C02 is in some way associated (possibly as sodium bicarbonate) with the plasma of the blood, and that the corpuscles have the power of acting like a weak acid, and of dissociating it from the soda, and thus raising its tension in the blood.
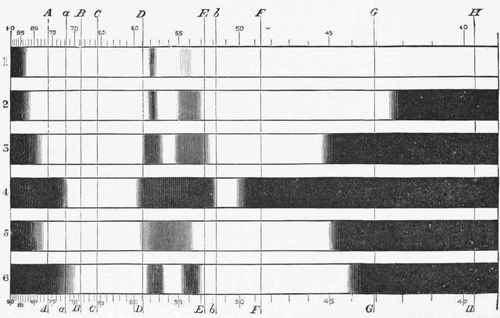
Fig. 155. - Spectra of Oxyhemoglobin, reduced haemoglobin, and CO-haemoglobin. {Gam-gee.) 1, 2, 3, and 4. Oxyhaemoglobin increasing in strength or thickness of solution. 5. Reduced haemoglobin. 6. CO-haemoglobin.
The great importance of the chemical nature of the union between the O and haemoglobin for external respiration becomes most striking when the actual manner in which the entrance of the O is effected is taken into account.
It must be remembered that the further we trace the air down the passages, the less will be the percentage of O found in it, and, therefore, a less pressure exerted by that gas. This is shown by the fact that the air given out by the latter half of a single expiration has less O and more CO2 than that of the first half. The most impure air lies in the alveoli of the lungs, for, since the tidal air scarcely fills the larger tubes, the air in the alveoli is only changed by diffusion with the impure air of the small bronchi. Any impediment to the ordinary ventilation of the alveoli so reduces the percentage, and, therefore, the tension of the O, that it would probably sink below that in the blood, and in that case, were it not a chemical union, the O would escape more readily from the blood in proportion as its tension in the blood exceeded that of the air of the alveoli. We know, however, that the blood retains a considerable quantity of oxygen even in the intense dyspnoea of suffocation.
In the same way the difference of tension of the C02 in the alveolar air and in the blood hardly explains the steady manner in which the C02 escapes, and it has, therefore, been suggested that this escape also depends in some way upon a chemical process, possibly connected with the union of the O and haemoglobin; because the admission of O to the blood seems to facilitate the exit of the C02.
The following table gives the approximate tension of the two gases in the different steps of the interchange in the case of dogs with a bronchial region occluded so that the air it contained could be examined. It shows that the tensions are such as to enable physical absorption to take some share in the entrance of the O as well as in the escape of the C02. A separate column gives the volumes per cent, of each gas, corresponding to these tensions as compared with the atmospheric standard. The physical process must occur before the oxygen and the haemoglobin meet, since the latter is bathed in the plasma, and further separated from the alveolar O by the vessel wall and epithelium.
co2 | 0 | |||
Tension in mm. Hg. | Corresponding Volume per cent. | Tension in mm. Hg. | Corresponding Volume per cent. | |
In arterial blood | 21. | 2.8 | 29.6 | 3.9 |
In venous blood | 41. | 5.4 | 22. | 2.9 |
In air of alveoli | 27. | 3.56 | 27.44 | 3.6 |
In expired air | 21 | 28 | 126.2 | 16.6 |
In atmosphere, . . . | 038 | 0.04 | 158. | 20.8 |
Continue to:


