Fermentation. Part 2
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Fermentation. Part 2
Yeast, torula cerevisiae or mycoderma vini, is a fungoid vegetable organism, composed of microscopic globules which attain a maximum diameter of about 1/4000 of an inch. Each globule is composed of a thin membranous cell wall having the composition of cellulose, C6 H10O5, and is filled with a gelatinous proteine compound, principally consisting of exceedingly minute germinal granules. There are two varieties of yeast, depending upon the mode of propagation.When fermentation is conducted below 45° F., the propagation is carried on by an increase of the germinal granules within the cells by assimilation of nutriment from the fermenting liquid, until the cell wall bursts and the partially organized granules which are liberated proceed in their development, forming in turn parent cells. Yeast formed in this manner is called by the German brewers bottom yeast {Unterhefe), and is the kind used in the fermentation of Bavarian beer. It is deposited during the process at the bottom of the fermenting tun in the form of a grayish viscid or gelatinous mass, the yeast being more or less mixed with other sedimentary matter.
When, however, the temperature at which the fermentation is conducted is much above 45°, say from 70° to 85°, the development of germinal matter becomes much more rapid, and it passes, according to Dr. Lionel Beale, through pores of the parent cell wall, perhaps carrying a thin pellicle with it, and makes its appearance upon the outside in the form of what have been called buds, first discovered by Cagniard de la Tour. (See fig. 1.) These buds, which for a time remain attached by pedicles to the parent cells, then go on developing, and in time become detached and assume the functions of parent cells; or buds may spring from them before their separation, and thus branches extending to some distance may be formed, as in fig. 2. Fig. 3 exhibits an appearance often noticed: several buds growing from one parent cell, each filled with granular matter, and presenting an irregular outline. On account of its branching structure, yeast which grows in this way becomes buoyant from the collection of hubbies of carbonic acid gas between the branches, and rises to the top of the liquid, and therefore is called top yeast (Oberhefe). It is the kind used in the fermentation of the wort of ale and strong spirituous liquors.
It would appear, however, that the mode of propagation does not instantly change in either variety upon a change of temperature. Thus top yeast placed in a fermentable liquid having a temperature of 45° is said not to pass into bottom yeast at once, as though a habit had been acquired; and conversely, bottom yeast will not develop as top yeast under a certain length of time. Yeast globules do not increase in number in pure sugar solutions, but the older globules waste away while the new buds grow at the expense of their contents. To effect the fermentation of 100 parts of sugar requires about one part of yeast, weighed when dry. When the proportion of sugar is greater the excess remains unaltered, the cells will bo ruptured, and the solution will be found to contain a certain quantity of lactate and acetate of ammonia, and other ammoniacal salts. When, however, instead of a pure sugar solution, a saccharine vegetable infusion, as sweet wort, is employed as the fermentable liquid, the yeast cells rapidly increase at the expense of the azotized matters which are present and which are essential as their nutriment. During the fermentation of beer they often increase to eight or ten times their original quantity.
The following table, according to Mitscherlich, gives the composition of yeast in its active and in its exhausted state, the amount of ash being deducted:
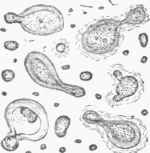
Fig. 1.-Growing yeast cells and most minute germs, magnified 2,000 diameters (Beale).
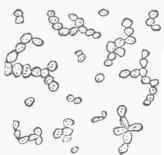
Fig. 2.-Yeast cells, growing during 4S hours, magnified 250 diameters (Beale).
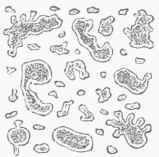
Fig. 3. - Growing yeast cells, showing diverticula, or buds, magnified 1,300 diameters (Beale).
CONSTITUENTS. | Active cells. | Spent cells. |
Carbon.................. | 47.0 | 47.6 |
Hydrogen............... | 6.6 | 7.2 |
Nitrogen.................... | 10.0 | 5.0 |
Oxygen................. | 35.8 | .... |
Sulphur....................... | 0.6 | ....... |
The inorganic matter represented by the ash amounts to about 7'5 per cent. of the dried yeast, and is composed, according to Mulder, entirely of phosphates of potash, soda, lime, and magnesia.-3. Acetic Fermentation. Liebig regarded the conversion of alcohol into acetic acid rather as a process of eremacausis, or slow oxidation, by which hydrogen was removed and oxygen substituted; but as the process is facilitated by ferments, particularly by the mycoder-ma aceti, it is generally regarded as a species of fermentation. Alcohol is readily oxidized by the influence of finely divided platinum into acetic acid, and also by binoxide of manganese and bichromate of potash. It is supposed that the reaction includes two stages: first the formation of aldehyde by the abstraction of two equivalents of hydrogen, water being at the same time formed; and subsequently the addition of one equivalent of oxygen, as represented by the following equations:
C2H6O | + | O | = | C2H4O | + | H2O |
Alcohol. | Aldehyde. | Water. | ||||
C2H4O | + | O | = | C2H4O2 |
Aldehyde. | Acetic acid. | |||
If the supply of oxygen be insufficient, much of the aldehyde remains unconverted into acetic acid, and on account of its great volatility may pass away in vapor. Pure diluted alcohol does not absorb oxygen from the air, but requires the presence of some inducing body which shall modify the atomic character of the oxygen, and also perhaps of that of the alcohol, so that the affinity of the constituent hydrogen and the atmospheric oxygen shall be increased. -4. Lactic Fermentation. When milk is left to stand for a time, the lactic sugar (C12H24O12) which it contains decomposes into lactic acid. The transformation is exceedingly simple, consisting merely in the splitting up of the molecules of sugar into a less complex arrangement, C12H24O12 becoming 2C3H6O3, or lactic acid. Caseine while passing into a state of decay was formerly supposed to be the ferment which induced the process; but according to Hallier and others, it consists of minute organisms which are developed from spores of penicillium crustaceum. (See figs. 4, 5, 6, 7.) The process is usually accompanied or immediately followed by the coagulation of the milk, an action which is generally ascrihed to the abstraction of the alkaline constituents of the caseine, which are supposed to hold it in solution; but it is asserted by some observers that coagulation of new milk by rennet often commences before any lactic acid makes its appearance.
Continue to:


