Galvanism, Or Voltaic Electricity. Part 10
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Galvanism, Or Voltaic Electricity. Part 10
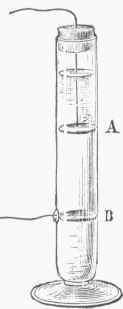
Fig. 23.
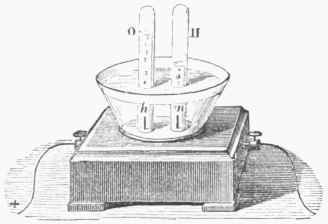
Fig. 24.-Voltameter.
- | A | A | + | |||
H2 | SO4 H2 | S04 H2 | S04 H2 | O | ||
+ | - + | i - + | - + | - |
Other binary compounds are very readily decomposed by electrolysis, as for instance iodide of potassium, KI, in this manner, the negative element of the compound molecule, iodine, going to the positive, and the positive element, potassium, going to the negative electrode. By means of a battery of 250 couples Sir Humphry Davy decomposed the alkalies potash and soda, showing them to be oxides of two hitherto unknown metals, potassium and sodium. The electrolysis of potash may be accomplished by placing a globule of mercury in a cavity made in a piece of caustic potash, which being moistened lies upon a piece of platinum foil. The latter is connected with the positive electrode of a Grove's or Bunsen's battery of six or seven cells, and the point of a wire, connected with the negative electrode, is placed in the mercury globule, which then attracts the positive element, metallic potassium, of the caustic potash, while the negative element, oxygen, is attracted by the plate of platinum. The potassium on being liberated forms with the mercury an amalgam, which on being subjected to distillation yields metallic potassium.
Any substance which is separated into component parts by electrolysis is called an electrolyte, and must be in a fluid condition so as to admit of displacement of the molecules. Although it is believed at the present time that the molecules of solid bodies do not touch each other, still it must of necessity be a condition that they are held by their polarity in such fixed relations that no external force is sufficient to release them until the attraction is diminished by heat or by some solvent. From the nature of molecular combination, one constituent of a molecule is electro-positive, and the other electro-negative; therefore, when subjected to the influence of electrical attraction, one of the constituents will be drawn to the positive and the other to the negative electrode of the battery. The positive electrode of a battery was named by Faraday the anode, and the negative the cathode, the former referring to the upward and the latter to the downward direction of the current. The elements or constituents of the electrolyte he called ions, the one going to the anode the anion, and the one going to the cathode the cation. Potassium, which is the most electropositive of all bodies, is therefore always a cation, and oxygen, the most electro-negative body, is always an anion.
As a general rule, the metals, or those constituents which contain them, are cations, while the non-metallic constituents are anions; but some elements may be sometimes cations and sometimes anions. In chloride of sodium, NaCl, for instance, chlorine is an anion, while in chloric acid, C12O5, it becomes a cation, or electro-positive element. In the electrolysis of ternary salts, which were formerly supposed to be constituted by the union of an acid with an oxide of a metal, it was held that there was a separation of this oxide from the acid, and then a subsequent separation of the oxygen and the metal, and their appearance respectively at the positive and negative electrodes; but this theory was only adapted to the one entertained with regard to the constitution of a salt. It is now held that ternary salts have in reality a binary constitution, the metal forming the electro-positive, and the salt radical the electro-negative constituent. The electrolysis of a salt may be shown by using a U-shaped tube as represented in fig. 25. A solution of the salt colored with sirup of violets is introduced into the tube, and the platinum electrodes of a battery are placed one in each leg.
After the current has passed for a time the fluid will have a red color about the positive, and a green color about the negative electrode, the red being caused by the action of the acid, and the green by that of the alkali, these bodies being formed subsequently to the electrolytic decomposition of the salt by the union of the metal with the oxygen of the water, and the union of the body SO4 with the hydrogen. It was the appearance of these acids and bases at the electrodes which led to the old theory of saline compounds. In electrolysis there is not merely a separation and recombination of constituents in the chain of molecules between the electrodes, but there is an actual passage of one to the positive and of the other to the negative electrode. This action was demonstrated by Davy in the following manner: A solution of sulphate of soda was placed in two vessels and connected by a bundle of asbestus moistened with the same solution. The electrodes of a battery being placed in the cups, it was after a time found that the cup connected with the positive electrode contained all the sulphuric acid, while the soda was all found in the other.
If one of the cups contains a solution of sulphate of soda, and the other a solution of chloride of sodium or of potassium, both metallic bases will after a time be found in the cup connected with the negative electrode, and the acid radicals in the other; and this transference of molecules will take place with any number of solutions, and through any number of cups which will not offer too great resistance to the passage of the current, provided that all the compounds which are formed by the interchange of molecules are soluble. If, however, an insoluble compound is formed by the union of any of the constituents in any of the vessels, it will be precipitated and thus finally eliminated from the solution. Thus if a solution of sulphate of potash is placed in A, fig. 26, connected with the negative electrode of a battery, distilled water in C, connected with the positive electrode, and a solution of caustic baryta in B, all being connected by threads of moistened asbestus, the passage of a current will effect no transference of sulphuric acid to the positive electrode, but a precipitate of sulphate of baryta will be formed in the cup B. The researches of Faraday have established the following laws of electrolysis: 1. Electrolysis cannot take place unless the electrolyte is a conductor.
Continue to:


