Acidimetry. Continued
Description
This section is from the "Encyclopedia Of Practical Receipts And Processes" book, by William B. Dick. Also available from Amazon: Dick's encyclopedia of practical receipts and processes.
Acidimetry. Continued
81. Table of Equivalents of Acids
81. Table of Equivalents of Acids. This table is based on the foregoing table of alkalis; so that, for instance, 1 equivalent (17 grains) of pure ammonia will exactly neutralize 1 equivalent (22 grains) dry carbonic acid, etc..
Grains.
Anhydrous acetic acid.............. 51
Hydrated or crystallized acetic acid... 60
Dry benzoic acid....................113
Crystallized benzoic acid............122
Dry boracic acid.................... 35
Crystallized boracic acid............. 62
Dry carbonic acid.................. 22
Dry citric acid...................... 58
Crystallized citric acid.............. 67
Dry hydrochloric acid............... 361/2
Liquid hydrochloric acid (sp.gr.1.16).. 109
Dry malic acid.....................58
Dry nitric acid...................... 54
Liquid nitric acid (sp. gr. 1.5)........ 67
" " " (sp. gr.1.42)...... 90
Dry oxalic acid.....................36
Crystallized oxalic acid.............. 63
Dry sulphuric acid.................. 40
Liquid sulphuric acid (sp. gr. 1.845... 49
Dry tartaric acid................... 66
Crystallized tartaric acid............75
82. Acidimeter
82. Acidimeter. An acidimeter is a glass tube, graduated with 100 divisions, each division representing 10 grains of distilled water, termed grain measures. The acidimeter is used for testing acids and alkalis, and is usually furnished with a lip for convenience in pouring by drops. Where great delicacy is required in pouring or dropping, various appliances are resorted to, by which the outward flow can be instantly arrested, merely by placing the finger or thumb on an orifice arranged for the ingress of air. In the illustrations, c denotes the place of egress for
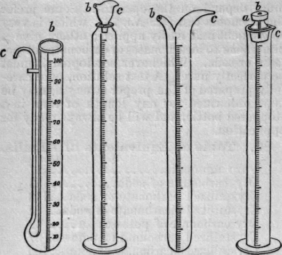
Fig. 1. Fig. 2. Fig. 3. Fig. 4.
the contained liquid; b, the orifice for the ingress of air, to be stopped by the finger or thumb; in Fig. 2, both orifices are in a hollow movable stopper; in Fig. 4, the air-hole only is in the stopper, a.
Fig. 1 represents Gay Lussac's Pouret.
Fig. 2, Normandy's modification of Schuster's Alkalimeter.
Fig. 3, Birck's Alkalimeter.
Fig. 4 is a simple acidimeter, with a stopper fitted to it, having a groove to correspond with the lip, and a vent-hole drilled through it to admit the air.
These modifications of the simple acidimeter are employed to allow of the test-liquid being added a single drop at a time, which is absolutely necessary during the first part of the process, to prevent undue effervescence, and consequent danger of loss of the liquid; and in the latter part it is equally indispensable in order to attain exact saturation. They dispense with the use of a separate pipette, being, in fact, acidimeters and pipettes combined.
Continue to:


