Chapter VIII. Theoretical Relations Between The Weight And Composition Of Distillate
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter VIII. Theoretical Relations Between The Weight And Composition Of Distillate
Application of Brown's Formula.1 - It has been pointed out in previous chapters1. That the vapour pressures of mixtures of two closely related compounds - and rarely of others - are represented with small error by
2. That when this formula holds good, the composition of the vapour from any mixture is given, approximately at any rate, by greatly from the mean ratio of the vapour pressures of the pure sub-stances at temperatures between their respective boiling points. We may conclude, then, that Brown's formula can be used without much error for mixtures of two closely related substances, and it is probable that the two formulae referred to above, when suitably modified, are applicable also to mixtures of three or more closely related substances.
Mixtures Of Two Components
Taking first the case of mixtures of two liquids, Brown's formula may be written where E = residue of liquid B at any instant, n = residue of liquid A at the same instant and dE; and dn =the weights of B and A, respectively, in the vapour.

Taking L and M as the weights of B and A originally present and we obtain by integration where y = quantity of the more volatile liquid B in unit weight of the distillate coming over at the instant when x is the quantity of liquid distilled. By means of this equation the changes of composition that take place in the course of a distillation may be traced, and the variation in the composition of the distillate represented graphically. To take a very simple case, suppose that c = 2 and that L = M = 1/2.
1 Barrell, Thomas and Young, " On the Separation of Three Liquids by Fractional Distillation," Phil. Mag., 1894, [V.], 37, 8.
In the diagram (Fig. 37), the amounts of distillate that have been collected are represented as abscissae, and the relative quantities of the two liquids, A and B, in the distillate as ordinates.
It will be seen that the composition of the distillate alters slowly at first, then more and more rapidly, also that while the first portion of the distillate contains a considerable amount of the less volatile substance, A, the last portion is very nearly free from the lower boiling component, B. These points are fully confirmed by experiment, and an explanation is afforded of the fact that it is much easier to separate the less volatile than the other component in a pure state.
By fractionating a few times in the ordinary way, collecting the distillates in six or eight fractions, we shall have a large excess of B in the first fraction and a still larger excess of A in the last.
Suppose, now, that two of these fractions, one containing B and A in the ratio of 9 : 1 and the other in the ratio 1 : 9, are distilled separately and completely ; the results will then be represented by Figs. 38 and 39. Again, it will be seen that the first tenth of the distillate from the first of these fractions is not so rich in B as the last tenth from the second fraction is in A ; the purification of A still proceeds more rapidly than that of B.

Fig. 37. -5=0-5, A =0.5, in original mixture.

Fig. 38. - B=0.9, 4 = 01, in mixture distilled.

Fig. 39.-5=0-1, A=0.9, in mixture distilled.
In making use of this formula it is assumed that no condensaticn (and therefore no fractionation) goes on in the still-head, but that the vapour reaches the condenser in the same state as when first evolved from the liquid in the still. By using an improved still-head (Chapters X. to XII.) a more rapid separation would be effected.
Mixtures Of Three Components
If we have a mixture of three closely related substances, C, B and A, it may be conjectured that the proportion of the three substances in the vapour at any instant is the same as that of the weights of the three substances in the residue in the still, each weight being multiplied by a suitable constant, which is approximately proportional to the vapour pressure of the corresponding liquid. Here again formulae may be obtained by integration, which enable us to follow the course of a distillation.
Taking the three constants as c = 4, 6 = 2, a = 1 - which are roughly proportional to the vapour pressures of methyl, ethyl and propyl acetates at the same temperature - and the original weights of the three liquids C, B and A as L = M = N = 1/3, Fig. 40 a represents the first distillation.
If the distillate were collected in five equal fractions they would have the following compositionTable 44
C or L. | B or M. | A or N. | |
Ha . | . 0.543 . | 0.300 . | 0.157 |
Il3 . . . | . 0.47 . | . 0.33 . | 0.20 |
IIy . . . | . 0.37 . | 0.365 . | 0.265 |
11 . . . | . 0.22 . | 0.39 . | 0.39 |
Iie . . . | . 0 047 . | 0.265 . | 0.687 |
It will be seen that while the last fraction is more than twice as rich in A as the original mixture and is nearly free from C, and the amount of C in the first fraction is 0.543, as against 0.333 in the original mixture, the fraction richest in B, the fourth, contains only 0.39 of that substance. It is evident, therefore, that the middle substance is the one that is the most difficult, and the least volatile sub-stance the one that is easiest to separate. If these five fractions were separately distilled we should get the results indicated in the curves, Fig. 40, b, c, d, e, f.
It is to be noted that in IIa, which is richest in the most volatile component, C, the amount of C rises from 0.543 to about 0.72 in the first fifth of the distillate. In IIe, which is richest in the least volatile liquid A, the amount of A rises from 0.687 to about 0.9 in the last fifth of the distillate. On the other hand, in 118, which is richest in B, the middle liquid, the improvement is merely from 0.39 to rather less than 0.45 in the third fifth of the distillate.

Fig. 40 (a to/). - Distillation of mixtures containing three components, A, B and C.
If the fractions richest in C, B and A respectively were redistilled we should have the following result for the best distillates :-
Improvement in C from . | 0.7 to 0.82 |
" B | 0.45 „ 0.50 |
A | 0.9 „ 0.98 |
Thus, the improvement m B continues to be very slow, and even when a fraction very rich in B is redistilled the improvement is very slight. Thus, on redistilling a mixture containing 0.02 of C, 0.96 of B and 002 of A, the amount of B advances only from 0.96 to 0.97 in the most favourable part of the distillate; it is thus clear that there must be far greater difficulty in separating the middle substance than either of the others from a mixture.
It is, however, not difficult to obtain fractions rich in C but free from A, and also rich in A but free from C, and the best method of obtaining B in a pure state is to carry out a series of preliminary fractionations in the manner described for methyl, ethyl and propyl acetates (p. 105) until all the fractions coming over below the boiling point of B contain only C and B, and all those coming over above that temperature contain only B and A. We have, then, only to deal with the separation of two components from the mixtures, and these separations will already be far advanced.
In 1902 Lord Rayleigh brought forward another theory of distillation. Denoting the weight of a given liquid mixture by w and the weight of the first component in it by y, y/w =E = the fractional amount of the first component in the liquid.
Calling the total weight of vapour dw and the weight of the first component in it dy, the fractional amount of the first component in the vapour
Therefore and if w0 and E0 denote the initial values of w and E


Lord Rayleigh makes the assumption that in mixtures containing very little of the first component the fraction of that component in the vapour is proportional to its fraction in the liquid, or and therefore

Rosanofl, Bacon and Schulze2 point out that Lord Rayleigh's theory is thus confined to very dilute mixtures, and that the solution of the problem proposed by Barrell, Thomas and Young is based on Brown's law, which is only of limited applicability. They themselves bring forward a more general theory based on the relationship expressed by their equation.1 which has been found to hold good in all types of cases.
1 Phil Mag., 1902, [5], 4, 527.
2 " A Theory of Fractional Distillation without Reflux Condensation," J. Amer. Chem. Soc, 1914, 36, 2000.
Let E and n denote the weights of the two components in the liquid and d£ and dn their weights in the vapour, pA and pB, as before, the partial pressures of the components, and m the molar fraction of the first component in the liquid. Then pA/pB, the ratio of the partial pressures, equals the ratio of the molar fractions in the vapour, the molar fractions being, of course, based on the actual molecular weights of the components. It is assumed that the molecular weights have the same value in the vapour as in the liquid, otherwise the thermodynamic equation of Duhem and Margules would not hold good. It may therefore be stated that the quotient of the ratios of the molar fractions in vapour and liquid is equal to the quotient of the ratios of the weights, that is to say -
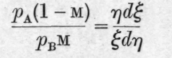
or

It follows from equation A that or therefore and all conclusions regarding the course of a fractional distillation based on this formula will be as reliable as equation A itself.
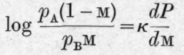
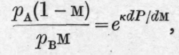
Denoting by £0 and m0 the initial weight and the initial molar fraction of the first component in the liquid the authors finally deduce the equation and for the second component, similarly

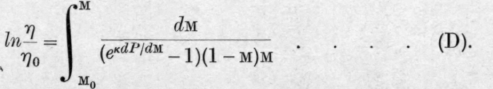
1 P. 93.
But as £ and 77 are connected by the equation where MB and MA are the molecular weights of B and A respectively, only one integration need be carried out.

The authors point out that equations C and D express the relation between the changes of weight and composition accompanying isothermal fractional distillation in terms of dP/dM, the slope of the total pressure curve.
When the total and partial pressure curves are straight lines, equation B becomes identical with Brown's formula, or and the integrals in C and D assume the values and

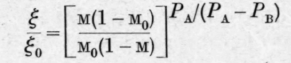

The authors have tested the theory, in the form of equations C and D, by direct experiment with satisfactory results.
See also W. K. Lewis, "Theory of Fractional Distillation," J. Ind. and Eng. Chem., 1909, 1, 522.
Continue to:


