Chapter XXII. Production Of Acetone And n-Butyl Alcohol By The Fermentation Process
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XXII. Production Of Acetone And n-Butyl Alcohol By The Fermentation Process
The production of n-butyl alcohol and other alcohols from carbohydrate material, directly by fermentation, has been known for a long time. The presence of iodoform-producing substances among the products of fermentation has been recorded occasionally. The first observation of acetone as the result of fermentation of carbohydrates was made by Schardinger,1 who obtained this substance together with acetic and formic acids. The isolation of an organism yielding acetone and n-butyl alcohol from amylaceous material was due to Fernbach and Strange.2 Since this initial discovery many other processes have been described which produce acetone and n-butyl or ethyl alcohol by the breakdown of carbohydrates under bacterial action.
Table 91 gives the yields of products obtained in the ordinary "acetone n-butyl alcohol " fermentation process.
In a normal fermentation the acidity of the mash increases from a very small initial value until a maximum is reached in from 13 to 17 hours after inoculation. (The length of time taken to reach this point is influenced by the percentage of inocu-lant used, temperature of the mash, etc.) When the maximum acidity is reached in 6.5 per cent maize mash, 3.5 to 4.5 c.c of N/10 alkali are required to neutralise 10 c.c. of mash. After this point is reached a very marked acceleration in the rate of production of acetone, n-butyl alcohol, carbon dioxide and hydrogen takes place. The acidity gradually falls to a constant value, 1.5 to 2.5 c.c. of N/10 alkali being required to neutralise 10 c.c. of mash (see Fig. 88, Curve 2).
1 Schardinger, Centr. Bakt., Part II., 1907, 18, 748.
2 Fembach and Strange, Eng. Patent, 1912, 21073.
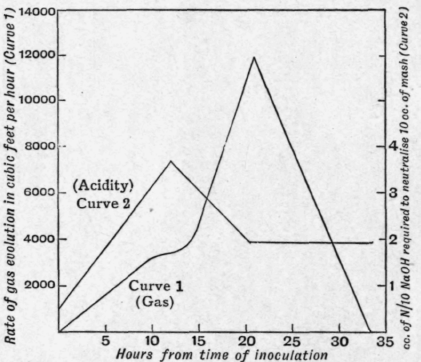
Fig. 88.
The rate of gas evolution is shown in Curve 1, Fig. 88, and the composition of the gas in Table 92.1
Table 91
1000 lb. maize containing 650 lb. starch. Volume of mash (6.5 per cent maize) = 1540 gallons.
lb. | Carbon content. | ||
650 lb. starch yield | 70 acetone 163 n-butyl alcohol | 43.5 105.7 | |
3410 cu. ft. of Co2 } evolved - at 27° and 760 mm. | = 390 C02 evolved | 106.3 | |
1 vol. of mash * at 38° dissolves 0.3 vol. C02. The latter being estimated at N.T P. | 74 cu. ft. C02 in sol. at 0° and 760} mm. | = 9.2 C02 in solution | 2.5 |
2090 cu. ft. H2 evolved at 27° and } 760 mm. Residual acidity | - 11 H2 = 12 "acids" containing | 5.7 | |
655.2 | 263.7 |
Theoretically 650 lb. of starch is equivalent to 722 lb. of hexose or 288.8 lb. of carbon.
* 1 vol. of distilled water at 38° dissolves 0.55 vols. C02 at N.T.P. where pressure of Co2=760 mm.-At 38° tension of water vapour = 49.3 mm. - Actual pressure due to gas = 710.7 mm. - as gas consists of 40 per cent H2 and 60 per cent C02 by vol., the partial pressure of the C02 = 426.4 mm. - At this pressure the solubility of the C02 is 0.31 vols, (at 0 and 760 mm. pressure) in 1 vol. of liquid. The fermentation liquors contain substances which influence the solubility and retention of carbon dioxide. Estimations of C02 in liquors from a series of fermentations gave results varying between 0.15 and 0.52 vols, of C02 dissolved in 1 vol. of mash liquor. A slightly lower value (0.3) than that of the solubility of C02 in water at 38° has been used.
The residual acidity is taken as acetic acid 56-5 per cent by weight, and butyric acid 43.5 per cent by weight. The non-volatile portion is recorded as butyric acid for the purpose of calculation.
Calculated as Percentage of Starch Fermented 100 g. starch gives 111.1 g. hexose and contains 44.4 g. carbon.
g. | Carbon. | |
100 g. starch gives | 10.77 acetone | 6.68 g. |
,, ,, | 25.07 n-butyl alcohol | 16.21 |
,, ,, | 61.41 carbon dioxide | 16.75 |
,, ,, | 1.60 hydrogen | - |
,, ,, | 1.80 residual acidity | 0.85 |
100.65 | 40.49 |
Rate of Gas Evolution, and Rise and Fall of Acidity throughout the Fermentation. - The curves in Fig. 88 show the changes in
(1) the rate of gas evolution, and
(2) the acidity during a fermentation. The figures are the: average of 12 fermentations of 6.5 per cent maize mash (40,000 gallons) with 4.7 per cent of inoculant.
The acidity is measured in c.c. of N/10 alkali required to neutralise 10 c c. of mash after boiling the solution to remove carbon dioxide. Phenolphthalein is used as indicator.
1 Reilly, Hickinbottom, Henley and Thaysen, Biochem. Journ., 1920, vol. xiv. No. 2, p. 229.
It will be noted that the rate of gas evolution rises steadily with the increase of acidity for some time ; it then becomes constant (in some cases it even slackens somewhat). As the acidity falls, the rate of gas evolution rises quickly to a maximum, and then falls rapidly until the end of the fermentation. Readings of the gas evolution were taken every hour. The acidity was estimated every three hours. The acidity at the end of the fermentation is generally higher than that of the mash when inoculated.
Continue to:


