Composition Of Liquid And Vapour Phases. Theoretical. Part 8
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Composition Of Liquid And Vapour Phases. Theoretical. Part 8
Oxygen And Nitrogen
Baly1 found that, for mixtures of liquid oxygen and nitrogen, Lehfeldt's formula gave very good results, but Brown's did not.
Associating Substances
As regards mixtures of ethyl alcohol, an associating substance, with benzene and with toluene, Lehfeldt points out that the relations between log t and log q are far from linear, and he did not attempt to find an equation for the curves.
Zawidski's Formula
Zawidski adopts the following equations to express the relations between the partial pressures, pt and p2, of the components in the mixture, the vapour pressures, P1 and P2, of the pure components, and the molecular fractional amounts, m and 1 - m, of the two substances in the mixture.
1 Baly, " On the Distillation of Liquid Air and the Composition of the Gaseous and Liquid Phases," Phil. Mag., 1900, [V.], 49, 517.
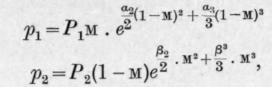
And where a2 and a3 are constants, the values of which can be ascertained from the partial pressure curves; or, by Margules's method, from the tangents to the total pressure curve at the extreme points where m = 0 and m = 1 by means of the equations:

Relation between Brown's and Zawidski's Formulae Zawidski points out that if, in his formula, the constants a and B vanish, the equations become p1 = P1m and p2 =P2(1 - m), whence p1/p2 =P1/P2 . M/(1-M) or P1/P2, = const. x m/(1 - m) ; and since, for substances of normal molecular weight, the partial pressures are proportional to the number of molecules present, the equation, in its final form, simply expresses Brown's law, taking the relative number of molecules instead of relative masses for both liquid and vapour.
Zawidski shows that this simple relation holds for mixtures of ethylene and propylene dibromides and of benzene and ethylene dichloride, but that for the first pair of liquids, the constant, 0.758, differs somewhat widely from the ratio P1/P2, 0.737, though the agreement is excellent for the second pair (0.880 and 0.881)
Zawidski shows, for these two pairs of substances, not only the correctness of the relation P = p1+p2 = P1M+P2 (1-M) which is equivalent to but also of the formulae p1 = P1M and p2 = P2(1-M) as will be seen from the diagram (Fig. 33) which is taken from his paper.

Fig. 33. Benzene and ethylene dichloride.
In the case of other mixtures of liquids with normal molecular weight, the constants a and B were found to have finite values, and the simple formulae p1 = P1M and p2 = P2(1 -m) were not found to be applicable ; the relations between the molecular composition and the pressures, whether total or partial, are, in fact, represented by curves. Thus, even with benzene and carbon tetrachloride the curvature, though slight, is unmistakable.
Benzene And Carbon Tetrachloride
For this pair of liquids Zawidski gives the constants a2 and a3, calculated by both methods; they are I. From the partial pressure curve, a2 = 0.308, a3 = 0.00733.
II. From the tangents (dtt/dM)0 = 90, (dTT/dM)l= -4.3, a2 = 0.312, a3= -0.0168.
Now the value of the tangents (dTT/dM)1 calculated from the first constants would be -5.0, so that in either case there is a negative value for the tangent when m = 1. If this is correct, there must be a mixture of maximum vapour pressure containing little benzene, and there is thus additional evidence (pp. 84 and 89) that these two liquids can form such a mixture, though the difference between the maximum pressure and the vapour pressure of carbon tetrachloride is probably too small to be determined by direct experiment.
For this and other pairs of liquids of normal molecular weight, the agreement between the observed molecular composition of the vapour and that derived from the calculated partial pressures is fairly good, though there are occasionally differences amounting to 4 or 5 per cent.
Carbon Disulphide And Methylal
In Table 34 below are given the results for carbon disulphide and methylal, so as to compare the calculated values with those obtained by means of Lehfeldt's formula.
Table 34
A = Carbon disulphide ; B = Methylal From {dTT/dM)0= + 578 and (dTTldM)1= - 1310 ; a'2 = 2.9 ; a.3= -1.89
Molecular percentage of Carbon disulphide. | |||||||
Liquid. | Vapour. | Liquid. | Vapour. | ||||
Observed. | Calculated. | | Observed. | Calculated. | | ||
496 | 8.98 | 8.95 | -0 03 | 60.60 | 54.76 | 53.97 | -0 79 |
1044 | 1739 | 17.09 | -0 30 | 68.03 | 59.21 | 57.87 | -1 34 |
1651 | 24.44 | 24.44 | 0 00 | 73.53 | 62.74 | 61.20 | -1.54 |
2719 | 34.39 | 34.45 | + 0 06 | 79.27 | 66.76 | 65.36 | -1.40 |
34.80 | 39.97 | 39 97 | 0.00 | 84.21 | 70.92 | 69.88 | -104 |
39.04 | 42.63 | 42.63 | 0.00 | 85.73 | 72.83 | 71.53 | -1.30 |
45.42 | 46.34 | 46.25 | -009 | 91.30 | 80.00 | 79.05 | -0 95 |
49.42 | 48.52 | 48.36 | -0.16 | 95.76 | 89.23 | 88.03 | -1.20 |
53.77 | 50.99 | 50.56 | -0 43 | ||||
The agreement up to 50 molecules per cent of carbon disulphide is excellent, but for mixtures richer in that component it is not nearly so satisfactory ; it is probable, however, that by altering the constants, better results might be obtained.
Continue to:


