Amino-Acetanilide From Nitro-Acetanilide
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Amino-Acetanilide From Nitro-Acetanilide
This azo component is prepared by the neutral reduction of nitro-acetanilde in practically the same way as has already been described several times. In an iron beaker provided with a propeller stirrer are placed 250 gms. cast-iron borings, 15 c.cs. 40 % acetic acid, and 500 c.cs. of water, the whole being then boiled vigorously for a few minutes, after which the moist nitro-acetanilide is added slowly in small portions with continuous stirring and boiling, so that the solution tested on filter paper remains colourless. When all has been added, boiling is continued for a further ten minutes, the evaporated water being replaced. After the liquid has cooled to 700 sufficient soda is added to give a perceptible alkaline reaction. The whole quantity obtained from 93 gms. aniline can easily be reduced in 20 minutes. If boiling be continued whilst neutralizing, or if too much soda be added, the nitro-acetanilide is easily hydrolysed. It is not possible, however, to precipitate at 700 all the iron which has gone into solution, and as this is absolutely necessary the remainder of the metal is precipitated with the minimum quantity of ammonium sulphide until a drop placed on filter paper gives no coloration with alkali sulphide. After this the mass may be filtered.
The solution, freed from iron and iron oxide, is now evaporated down to 400 c.cs. over a bare flame. On cooling, the aminoca. 10 gms.
2 50 gms. Fe. 15 c.cs.
(40 %). Acetic acid.
acetanilide separates out in beautiful long needles. The yield from 93 gms. aniline is about 75 gms. pure base. The mother-liquor, which always contains about 15 % of less pure products, is evaporated down further after standing for a day, and is then again allowed to crystallize.
The product so obtained is sufficiently pure for the works, but if desired absolutely pure it may be recrystallized from a little water, preferably with the addition of animal charcoal. The solutions, which on the large scale are evaporated down in vacuo, yield a purer amino-acetanilide.
On hydrolysing the amino-acetanilide, exactly as in the case of mtraniline, the important p-phenylene-diamine is obtained. It is extremely easily oxidized and is therefore hydrolysed either in complete absence of air, or by boiling with dilute sulphuric acid.
Notes on Works Technique and Practice. - p-Nitraniline is one of the most important products of the aniline dye industry. It serves not only for the preparation of solid colours in powder form, but also, and to a still greater extent, for the production of Para Red, which is an azo combination formed on the cotton fibre with β-naphthol. The nitraniline is prepared by two different processes; the most important is that starting from aniline. The manufacture of acetanilide is carried out either in enamel-lined or in aluminium vessels. The mother-liquors are worked up for sodium acetate. The other process starts from p-nitro-chlorbenzene, which is converted into nitraniline on heating in an autoclave with ammonia:l

Since very high pressures are produced on heating to 200 , many factories fear to use this process, although it gives a p-nitraniline which is much purer and just as cheap. For especially pure Para Red, many dyers prefer nitraniline made by this method, which is carried out successfully in certain of the smaller works.
1 See D. R. P. 148749.
Ortho- and Para-Nitrophenol and their Alkyl Ethers.
Reaction:
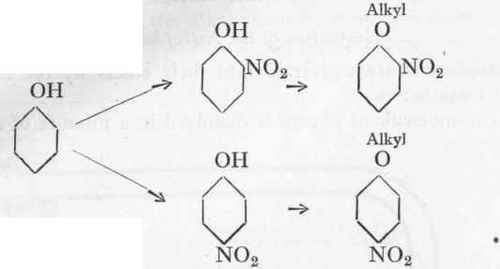
93 Gms. Phenol are melted with 20 c.cs. water, and the liquid mixture is allowed to drip into a solution of 150 gms. sodium nitrate in 400 c.cs. of water and 250 gms. concentrated sulphuric acid. During the addition the liquid must be kept well stirred and the temperature below 200. After all has been added, stirring is continued for a further 2 hours. The mother-liquor is then poured off the resinous mixture of the nitro bodies, and the residue melted with 500 c.cs. of water with the addition of sufficient chalk to give a completely neutral reaction with litmus. The wash-liquor is thrown away and the washing repeated. The crude nitrophenol freed from nitric acid is now steam-distilled, using a wide condenser. About 40 gms. of pure o-nitrophenol pass over. The residue left in the flask is then allowed to cool down, and after standing for 24 hours is filtered off from the mother-liquor. The residue is boiled up with 1 litre of 2 % hydrochloric acid and filtered through a folded filter. The pure p-nitrophenol crystallizes out from the hot solution in long, practically white needles; if necessary, the extraction may be repeated.
The yield is about 40 gms. ortho- and about the same quantity of para-derivative. Treatment of the crude nitrophenol with caustic soda solution has a very harmful effect, although given in various recipes, as the lye has an immediate resinifying action.
Notes on Works Technique and Practice. - In carrying out the distillation on the large scale, either worm condensers standing in warm water, or straight condensers fed with warm water, are used, in order to prevent "freezing up." Preferably the steam is heated to 1100 as very little is then required.
0- and p-Nitrophenols are the starting-point for the preparation of o- and p-phenetidine, and for o- and p-anisidine. From o-nitroanisol, further, dianisidine is prepared, which gives the finest direct blue on the market (Diamine Pure Blue, Chicago Blue, etc.).
93 gms. Phenol. 20 c.cs. H2O. 150 gms. NaNo3. 250 gms. H2So4, 66° Be. 400 c.cs. H2O. CaCo3.
ca. 1000 c.cs. 2 % Hc1.
Alkylation of the Nitrophenols.
Nitrophenols are converted into their ethers by the following general method:-
1 Gm.-molecule of phenol is dissolved in a mixture of 400 c.cs.
Continue to:


