Preparation Of An Amino-Naphthol Sulphonic Acid From The Corre Sponding Hydroxy-Nitroso Compound (Quinone Monoxime)
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Preparation Of An Amino-Naphthol Sulphonic Acid From The Corre Sponding Hydroxy-Nitroso Compound (Quinone Monoxime)
1:2:4-Amino-naphthol Sulphonic Acid from β-Naphthol.
1. Nitroso-β-naphthol. - 1oo gms. of β-naphthol1 are dissolved in 90 gms. of 35 % caustic soda lye and 1 litre of water at 500, contained in a glass jar of 3 litres capacity. To this solution, which should react faintly but distinctly to thiazole paper, 50 gms. of 100 % sodium nitrite are added, and the mixture made up with water and ice to 2 litres, temperature 0°. About 220 gms. of 40 % sulphuric acid are then allowed to run into this with good stirring during 3 hours; at the end of the time the solution should be distinctly acid to Congo and should react with nitrite paper. After
100 gms. β-Naphthol.
90 gms. NaOH
(35 %)
1 litre H2O.
50 gms. NaNO,.
220 gms. H2So4 (40 %).
1 A gm.-molecule is not taken a! this would need too great a volume of liquid.
10 hours the nitroso-naphthol is filtered off on a big porcelain filter and is well washed. It is chemically pure, assuming that the original β-naphthol was also pure.
2. Reduction and Conversion into the Amino-naphthol Sulphonic Acid. - The still moist nitroso-naphthol is stirred up with a little water in a glass jar and cooled down to 5o with ice. To the homogeneous paste is added quickly 260 gms. sodium bisulphite solution (about 25 % So2). The nitroso-naphthol goes into solution after a short time, a further small quantity of dilute caustic soda being cautiously added if necessary.
The solution contains some resinous constituents, and is therefore filtered. (By salting out the hydroxylamine sulphonic acid which is formed, Alsace Green N or Dioxine N of commerce is obtained, a dye which plays a certain part in calico-printing, the iron lake being very fast to light.)
The volume of the filtered solution is about 1 1/4 litres. It is placed in a jar and is then treated at 250 with 100 gms. sulphuric acid (66°Be.) which has been diluted with 200 gms. of water. The solution at the end should show a strongly mineral acid reaction. After 1 hour it is warmed to 500 and allowed to stand overnight; the contents of the jar solidify to a solid cake of free amino-naphthol sulphonic acid. This is filtered off and thoroughly washed out with water. The yield is about 90 %, calculated on the β-naphthol used.
260 gms. Bisulphite.
(25 %)
100 gms. H2So4, 66° Be. 200 gms. H2O.
Reaction:

Quinone oxime.

"Dioxine."
Amino-naphthol sulphonic acid 1:2:4.
Amino-naphthols of this type cannot be diazotized by the method used for other amines since, on treating with mineral acids and sodium nitrite, quinones are formed and only traces of the desired diazo compound. These diazo bodies, however, may be obtained quantitatively by treating the free acid (as obtained after filtering off and washing) in concentrated suspension with nitrite in presence of a molecule of zinc chloride or of a very small quantity of a copper salt. Both methods are employed, the patent literature giving the essential details (D. R. P. 171024 G.).
Amino-naphthol sulphonic acid 1:2:4 is an intermediate product for important ortho-hydroxyazo colours which were discovered simultaneously by Kalle, Geigy, and the B.A.S.F. When coupled with naphthols they yield very fast blue-black chrome dyes; the nitro derivatives (Sandmeyer-Hagenbach) are the cheapest chrome blacks for wool on the market, and have scarcely been surpassed for fastness (Erio Chrome Black T and A).
It is also of interest to notice that those amines which are substituted in the di-ortho positions couple with a-naphthol in the ortho-position to the hydroxyl. Thus from 1:24-amino-naphthol sulphonic acid, or, rather, from its diazo compound and strongly alkaline α-naphthol solution, the ortho-hydroxyazo compound is produced quantitatively (Erio Chrome Blue-Black B, Geigy). These diazo compounds are so stable that they can easily be nitrated in sulphuric acid solution by means of "mixed acid "; the nitro diazo compounds yield the Chrome Blacks mentioned above. Many 2:6 di-substituted anilines also couple with α-naphthol in the ortho position yielding products fast to alkali, whilst from ortho-hydroxy diazo compounds and α-naphthol chromable azo colours are formed.
The method of sulphonation by means of sulphurous acid mentioned above, is also applied to the preparation of amino-phenol disulphonic acid from nitroso-dimethyl-aniline and sodium bisulphite. During the conversion to the disulphonic acid, the dimethyl-amino group is split off leading to the formation of the p-amino-phenol derivative.
Reaction:
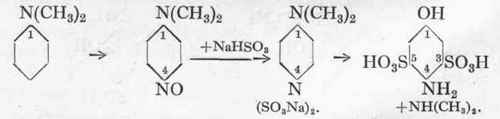
Pure dimethylamine is produced at the same time, which is a commercial product.
Fig. 12. - Separating funnel and extraction vessel provided with propeller-stirrer and sight-glass (scale 1 .30).
i

Fig. 13. - Centrifuge with underneath drive (scale 1 :3o).
Plate V.
Continue to:


