Roasting. Continued
Description
This section is from the book "Leaching Gold and Silver Ores. The Plattner And Kiss Processes: A Practical Treatise", by Charles Howard Aaron. Also available from Amazon: Leaching Gold And Silver Ores.
Roasting. Continued
Chloridizing Roast
20. Chloridizing Roast. At the commencement this is the same as an oxidizing roast; but salt is mixed with the ore, either at the time of charging the furnace or at a certain stage of the operation. The quantity of salt used depends on circumstances, and varies from one to twenty per cent. of the weight of the ore.
21. Salt is a compound of the metal sodium with chlorine, and is the cheapest source of chlorine. The chlorine of the salt is transferred, under the action of heat, from the sodium to the other metals, by a variety of agencies, chiefly by means of sulphur and oxygen, for which the sodium has a greater affinity, whence it happens that, when another metal sulphate is heated in contact with sodium chloride, an exchange takes place, the sodium takes the sulphur and oxygen, and forms sodium sulphate; the other metal takes the chlorine, and forms a chloride. In this way, iron, copper, zinc, lead, and silver sulphates form chlorides, while a corresponding proportion of salt forms sodium sulphate. For this reason, sulphur is necessary in a chloridizing roast, since without it sulphates cannot exist.
22. The higher sulphur oxide, formerly called dry sulphuric acid, but now known by the name of sulphuric anhydride, also plays a prominent part in the decomposition of the salt and the evolution of chlorine. The latter acts upon any remaining sulphides, and to some extent on oxides, converting them into chlorides. It also acts on metallic silver.
23. Among other agencies involved in the formation of metal chlorides, is that of steam, from burning fuel. Steam, in contact with salt and quartz, at a red heat, produces hydrochloric acid, which assists in the work. It also decomposes some of the base chlorides, especially the volatile ones, thus giving the silver the benefit of their chlorine. For this reason, steam is sometimes, in the case of rich ores, admitted to the roasting chamber by means of a perforated pipe laid in the firewall, which is made hollow, with openings on the side next to the ore. This, however, causes an increased consumption of fuel.
24. As the heat still increases, the base metal chlorides are decomposed, just as the sulphates are decomposed in the dead roast. They give off the whole, or a part of their chlorine, remaining, or volatilizing, as chlorides of a lower degree, or taking oxygen from the air and becoming oxychlorides, or oxides. Silver chloride is not decomposed by heat.
25. Iron perchloride, formed quite early in the roasting, is volatile, and some of it flies away, while another part, giving off chlorine, is reduced to proto-chloride, and this again, losing the remaining chlorine, takes oxygen from the air, and forms iron peroxide, which remains in the ore.
26. Copper protochloride gives off half its chlorine, becoming dichloride, which volatilizes to some extent, imparting a deep blue color to the flames.
27. Lead sulphate requires a rather high heat to convert it into chloride, so that it remains in part, sometimes wholly, unchanged. The chloride gives off some chlorine and takes oxygen, becoming oxychloride.
28. Zinc volatilizes partly, in some form, and produces in the flues hard concretions of oxide, or carbonate, which must be removed from time to time. The oxide is quite stable.
29. Antimony and arsenic form volatile chlorides as well as oxides, which, to a great extent, go up the chimney.
30. Gold forms, at a very low heat, a peculiar chloride which decomposes at a higher temperature, and then remains metallic, except as a portion of it may be again chloridized, in the same form, by chlorine from decomposing base chlorides, during the cooling of the ore, after being withdrawn from the furnace. The gold chloride formed in roasting contains less chlorine than that formed by the cold gas, and is not soluble in water, but dissolves in the solvent used for the silver leaching. The formation of this compound cannot be relied on as a process for extracting gold.
31. The presence of lead is disadvantageous for the roasting, because its compounds melt too easily, and because the oxide and chloride volatilize, to the injury of the workman's health; and for the leaching, because the sulphate and chloride are soluble in the silver leach, and the sulphate cannot be removed from the ore by washing with water; as, however, the chloride is soluble in hot water, it is preferable to the sulphate, as it can be removed from the ore before the silver extraction begins.
32. Silver chloride is not very volatile by itself, but in some cases becomes so, apparently from the influence of base metal chlorides, notably that of iron perchlo-ride, when too much heat is used early in the roasting. Antimony and zinc also tend to cause volatilization of silver; so as it does not require a very high heat to form the silver chloride, the roasting is conducted, not only with a very low heat at the beginning, but without an extremely high temperature at any time, and even a moderately high degree is maintained for a short time only towards the end. A chloridizing roast is a roast which, beginning with oxidation, ends by leaving, as nearly as possible, all the silver in the form of chloride in the ore.
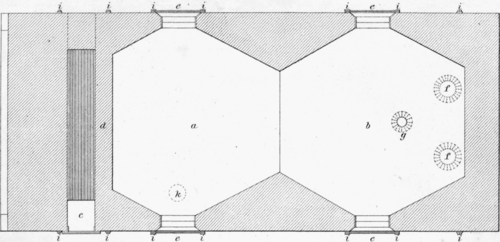
Scale; 1/4 inch = 1 foot. a.First hearth. b. Second hearth. c.Fire place. d.Firewall. e.Doors. f.Flues. g.Chargehole. i.Buckstay. k.Discharge hole.
Plate I.
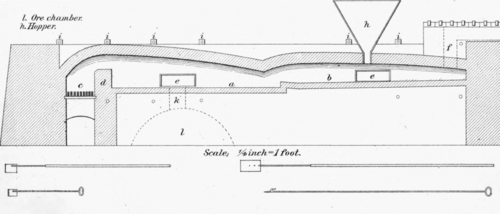
Plate II.
Continue to:


