Measuring Electromotive Force
Description
This section is from "Scientific American Supplement Volumes 275, 286, 288, 299, 303, 312, 315, 324, 344 and 358". Also available from Amazon: Scientific American Reference Book.
Measuring Electromotive Force
Coulomb's torsion balance has been adapted by M. Baille to the measurement of low electromotive forces in a very successful manner, and has been found preferable by him to the delicate electrometers of Sir W. Thomson. It is necessary to guard it from disturbances due to extraneous electric influences and the trembling of the ground. These can be eliminated completely by encircling the instrument in a metal case connected to earth, and mounting it on solid pillars in a still place. Heat also has a disturbing effect, and makes itself felt in the torsion of the fiber and the cage surrounding the lever. These effects are warded off by inclosing the instrument in a non-conducting jacket of wood shavings.
The apparatus of M. Baille consists of an annealed silver torsion wire of 2.70 meters long, and a lever 0.50 meter long, carrying at each extremity a ball of copper, gilded, and three centimeters in diameter. Similar balls are fixed at the corners of a square 20.5 meters in the side, and connected in diagonal pairs by fine wire. The lever placed at equal distances from the fixed balls communicates, by the medium of the torsion wire, with the positive pole of a battery, P, the other pole being to earth.
Owing to some unaccountable variations in the change of the lever or needle, M. Baille was obliged to measure the change at each observation. This was done by joining the + pole of the battery to the needle, and one pair of the fixed balls, and observing the deflection; then the deflection produced by the other balls was observed. This operation was repeated several times.
The battery, X, to be measured consisted of ten similar elements, and one pole of it was connected to the fixed balls, while the other pole was connected to the earth. The needle, of course, remained in contact with the + pole of the charging battery, P.
The deflections were read from a clear glass scale, placed at a distance of 3.30 meters from the needle, and the results worked out from Coulomb's static formula,
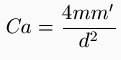 , with
, with

In M. Baillie's experiments, O = 437³, and Σpr²= 32171.6 (centimeter grammes), the needle having been constructed of a geometrical form.
The following numbers represent the potential of an element of the battery--that is to say, the quantity of electricity that the pole of that battery spreads upon a sphere of one centimeter radius. They are expressed in units of electricity, the unit being the quantity of electricity which, acting upon a similar unit at a distance of one centimeter, produces a repulsion equal to one gramme:
Volta pile 0.03415 open circuit. Zinc, sulphate of copper, copper 0.02997 " Zinc, acidulated water, copper, sulphate of copper 0.03709 " Zinc, salt water, carbon peroxide of manganese 0.05282 " Zinc, salt water, platinum, chloride of platinum 0.05027 " Zinc, acidulated water, carbon nitric acid 0.06285 "
These results were obtained just upon charging the batteries, and are, therefore, slightly higher than the potentials given after the batteries became older. The sulphate of copper cells kept about their maximum value longest, but they showed variations of about 10 per cent.
Continue to:


