Bunte's Burette For The Analysis Of Furnace Gases
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
Bunte's Burette For The Analysis Of Furnace Gases
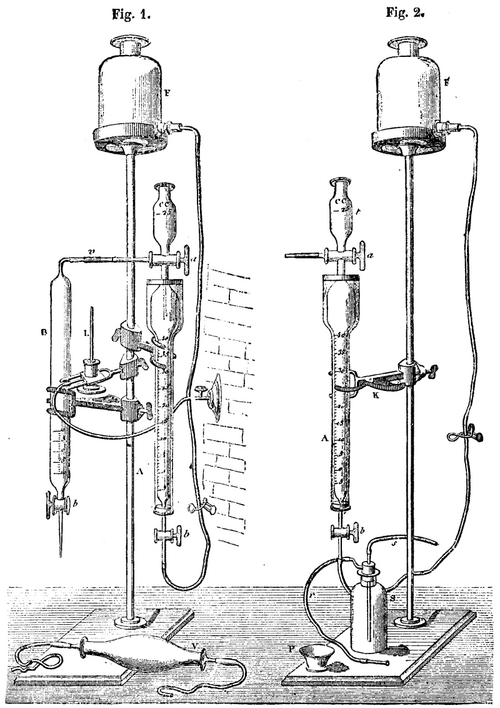
For analyzing the gases of blast-furnaces the various apparatus of Orsat have long been employed; but, by reason of its simplicity, the burette devised by Dr. Bünte, and shown in the accompanying figures, is much easier to use. Besides, it permits of a much better and more rapid absorption of the oxide of carbon; and yet, for the lost fractions of the latter, it is necessary to replace a part of the absorbing liquid three or four times. The absorbing liquid is prepared by making a saturated solution of chloride of copper in hydrochloric acid, and adding thereto a small quantity of dissolved chloride of tin. Afterward, there are added to the decanted mixture a few spirals of red copper, and the mixture is then carefully kept from contact with the air.
To fill the burette with gas, the three-way cock, a, is so placed that the axial aperture shall be in communication with the graduated part, A, of the burette. After this, water is poured into the funnel, t, and the burette is put in communication with the gas reservoir by means of a rubber tube. The lower point of the burette is put in communication with a rubber pump, V (Fig. 2), on an aspirator (the cock, b, being left open), and the gas is sucked in until all the air that was in the apparatus has been expelled from it. The cocks, a and b, are turned 90 degrees. The water in the funnel prevents the gases communicating with the top. The point of the three-way cock is afterward closed with a rubber tube and glass rod.
If the gas happens to be in the reservoir of an aspirator, it is made to pass into the apparatus in the following manner: The burette is completely filled with water, and the point of the three-way cock is put in communication with a reservoir. If the gas is under pressure, a portion of it is allowed to escape through the capillary tube into the water in the funnel, by turning the cock, a, properly, and thus all the water in the conduit is entirely expelled. Afterward a is turned 180°, and the lower cock, b, is opened. While the water is flowing through b, the burette becomes filled with gas.
Mode Of Measuring The Gases And Absorption
The tube that communicates with the vessel, F, is put in communication, after the latter has been completely filled with water, with the point of the cock, b (Fig. 2). Then the latter is opened, as is also the pinch cock on the rubber tubing, and water is allowed to enter the burette through the bottom until the level is at the zero of the graduation. There are then 100 cubic centimeters in the burette. The superfluous gas has escaped through the cock, a, and passed through the water in the funnel. The cock, a, is afterward closed by turning it 90°. To cause the absorbing liquid to pass into the burette, the water in the graduated cylinder is made to flow by connecting the rubber tube, s, of the bottle, S, with the point of the burette. The cock is opened, and suction is effected with the mouth of the tube, r. When the water has flowed out to nearly the last drop, b is closed and the suction bottle is removed. The absorbing liquid (caustic potassa or pyrogallate of potassa) is poured into a porcelain capsule, P, and the point of the burette is dipped into the liquid. If the cock, b, be opened, the absorbing liquid will be sucked into the burette.
In order to hasten the absorption, the cock, b, is closed, and the burette is shaken horizontally, the aperture of the funnel being closed by the hand during the operation.
If not enough absorbing liquid has entered, there may be sucked into the burette, by the process described above, a new quantity of liquid. The reaction finished, the graduated cylinder is put in communication with the funnel by turning the cock, a. The water is allowed to run from the funnel, and the latter is filled again with water up to the mark. The gas is then again under the same pressure as at the beginning.
After the level has become constant, the quantity of gas remaining is measured. The contraction that has taken place gives, in hundredths of the total volume, the volume of the gas absorbed.
When it is desired to make an analysis of smoke due to combustion, caustic potassa is first sucked into the burette. After complete absorption, and after putting the gas at the same pressure, the diminution gives the volume of carbonic acid.
To determine the oxygen in the remaining gas, a portion of the caustic potash is allowed to flow out, and an aqueous solution of pyrogallic acid and potash is allowed to enter. The presence of oxygen is revealed by the color of the liquid, which becomes darker.
The gas is then agitated with the absorbing liquid until, upon opening the cock, a, the liquid remains in the capillary tube, that is to say, until no more water runs from the funnel into the burette. To make a quantitative analysis of the carbon contained in gas, the pyrogallate of potash must be entirely removed from the burette. To do this, the liquid is sucked out by means of the flask, S, until there remain only a few drops; then the cock, a, is opened and water is allowed to flow from the funnel along the sides of the burette. Then a is closed, and the washing water is sucked in the same manner. By repeating this manipulation several times, the absorbing liquid is completely removed. The acid solution of chloride of copper is then allowed to enter.
As the absorbing liquids adhere to the glass, it is better, before noting the level, to replace these liquids by water. The cocks, a and b, are opened, and water is allowed to enter from the funnel, the absorbing liquid being made to flow at the same time through the cock, b.
When an acid solution of chloride of copper is employed, dilute hydrochloric acid is used instead of water.
Fig. 2 shows the arrangement of the apparatus for the quantitative analysis of oxide of carbon and hydrogen by combustion. The gas in the burette is first mixed with atmospheric air, by allowing the liquid to flow through b, and causing air to enter through the axial aperture of the three way cock, a, after cutting off communication at v. Then, as shown in the figure, the burette is connected with the tube, B, which is filled with water up to the narrow curved part, and the interior of the burette is made to communicate with the combustion tube, v, by turning the cock, a. The combustion tube is heated by means of a Bunsen burner or alcohol lamp, L. It is necessary to proceed, so that all the water shall be driven from the cock and the capillary tube, and that it shall be sent into the burette. The combustion is effected by causing the mixture of gas to pass from the burette into the tube, B, through the tube, v, heated to redness, into which there passes a palladium wire. Water is allowed to flow through the point of the tube, B, while from the flask, F, it enters through the bottom into the burette, so as to drive out the gas.
The water is allowed to rise into the burette as far as the cock, and the cocks, b and b¹, are afterward closed.
DR. BÜNTE'S GAS BURETTE
By a contrary operation, the gas is made to pass from B into the burette. It is then allowed to cool, and, after the pressure has been established again, the contraction is measured. If the gas burned is hydrogen, the contraction multiplied by two-thirds gives the original volume of the hydrogen gas burned. If the gas burned is oxide of carbon, there forms an equal volume of carbonic acid, and the contraction is the half of CO. Thus, to analyze CO, a portion of the liquid is removed from the burette, then caustic potash is allowed to enter, and the process goes on as explained above.
The total contraction resulting from combustion and absorption, multiplied by two-thirds, gives the volume of the oxide of carbon.
The hydrogen and oxide carbon may thus be quantitatively analyzed together or separately.--Revue Industrielle.
Continue to:


