Scrivanow's Chloride Of Silver Pile
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
Scrivanow's Chloride Of Silver Pile
Considerable attention has been attracted lately at Paris among those who are interested in electrical novelties to a chloride of silver pile invented by Mr. Scrivanow. The experiments to which it has been submitted are, in some respects, sufficiently extraordinary to cause us to make them known to our readers, along with the inventor's description of the apparatus.
Mr. Scrivanow's intention appears to be to apply this pile to the lighting of apartments, and even to the running of small motors, and, for the purpose of actuating sewing machines, he has already constructed a small model whose external dimensions are 160 x 100 x 90 millimeters.
"My invention," says the inventor, "is intended as an electric pile capable of regeneration. The annexed Fig. 1 shows a vertical arrangement of the apparatus, and Fig. 2 a horizontal one. In the latter, two elements are represented superposed.
"My pile consists of a prism of retort carbon (a) covered on every side with pure chloride of silver (b). The carbon thus prepared is immersed in a solution of hydrate of potassium (KHO) or of hydrate of sodium (NaHO), marking 1.30 to 1.45 by the Baumé areometer, the solvent being water.
"In the vicinity of the carbon is arranged the plate to be attacked--a plate of zinc (c) of good quality. The surface of the electrodes, and their distance apart, depends upon the effects that it is desired to obtain, and is determined in accordance with the well known principles of electro-kinetics.
"The chemical reactions that take place in this couple are multiple. In contact with a sufficiently concentrated solution of hydrate of potassium or sodium, the chloride of silver, especially if it has been recently prepared, passes partially into the state of brown or black oxide, so that the carbon becomes covered, after remaining sufficiently long in the exciting liquid, with a mixture of chloride and oxide of silver. When the circuit is closed, the chloride becomes reduced to a spongy metallic state and adheres to the surface of the carbon. At the same time the zinc passes, in the alkaline solution, into a state of chloride and of soluble combination of zinc oxide and of alkali.
"To avoid all loss of silver I cover the carbon with asbestos paper, or with cloth of the same material, d. My piles are arranged in ebonite vessels, A, which are flat, as in Fig. 1, or round, as in Fig. 2.
"In Fig 1 there is seen, at e, gutta-percha separating the zinc from the carbon at the base.
"Under such conditions, we obtain a powerful couple that possesses an electro-motive power of 1.5 to 1.8 volts, according to the concentration of the exciting liquid. The internal resistance is extremely feeble. I have obtained with piles arranged like those shown in the figures nearly 0.06 ohm, the measurements having been taken from a newly charged pile.
"When the element is used up, and, notably, when all the chloride of silver is reduced, it is only necessary to plunge the carbon with its asbestos covering (after washing it in water) into a chloridizing bath, in order to bring back the metallic silver that invests the carbon to a state of chloride, and thus restore the pile to its primitive energy. After this the carbon is washed and put back into the exciting liquid.
"These reductions of the chloride of silver during the operation of the pile can be reproduced ad infinitum, since they are accompanied by no loss of metal. The alkaline liquid is sufficient in quantity for two successive charges of the couple.
"The chloridizing bath consists of 100 parts of acetic acid, 5 to 6 parts, by weight, of hydrochloric acid, and about 30 parts of water.
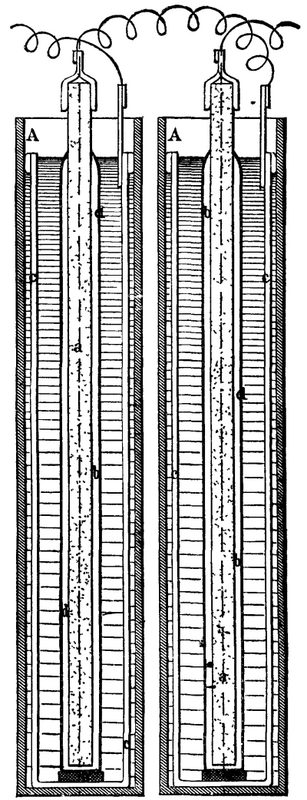
FIG. 1.--SCRIVANOW'S CHLORIDE OF SILVER PILE.
"Other acids may be employed equally as well. A bath composed of chlorochromate of potassium and nitric or sulphuric acid makes an excellent regenerator.
"To sum up, I claim as the distinctive characters of my pile:
"1. The use of the potassic or sodic alkaline liquid conjointly with chloride of silver, and the oxide of the same, that forms through the immersion of the carbon in a chloridizing bath.
"2. The use of retort or other carbon covered with the salt of silver above specified.
"3. The arrangement and construction of my pile as I have described."
In the experiments recently tried with Mr. Scrivanow's pile, a large sized battery was made use of, whose dimensions were 300 x 145 x 125 millimeters, and whose weight was from 5 to 6 kilogrammes. The results were: intensity, 1 ampere; electro-motive power, 25 volts, corresponding to an energy of 25 volt-amperes, or about 2.5 kilogrammeters per second. The pile was covered with a copper jacket whose upper parts supported two Swan lamps. Upon putting on the cover a contact was formed with the electrodes, and it was possible by means of a commutator key with three eccentrics to light or extinguish one of the lamps or both at once. A single element would have sufficed to keep one Swan lamp of feeble resistance lighted for 20 hours. Accepting the data given above and the 20 hours' uninterrupted duration of the pile's operation the power furnished by this large model is equal to 2.5 x 20 x 3,600 = 180,000 kilogrammeters.
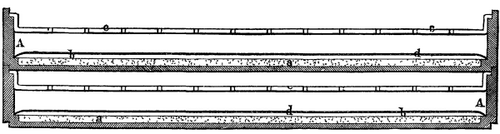
FIG. 1.--SCRIVANOW'S CHLORIDE OF SILVER PILE.
In our opinion, Mr. Scrivanow's pile is not adapted for industrial use because of the expense of the silver and the frequent manipulations it requires, but it has the advantage, however, of possessing, along with its small size and little weight, a disposable energy of from 150,000 to 200,000 kilogrammeters utilizable at the will of the consumer and securing to him a certain number of applications, either for lighting or the production of power. It appears to us to be specially destined to become a rival to the bichromate of potash pile for actuating electric motors applied to the directing of balloons.--Revue Industrielle.
Continue to:


