Further Researches Upon The Element Fluorine
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
Further Researches Upon The Element Fluorine
By A.E. TUTTON.
Since the publication by M. Moissan of his celebrated paper in the Annales de Chimîe et de Physique for December, 1887, describing the manner in which he had succeeded in isolating this remarkable gaseous element, a considerable amount of additional information has been acquired concerning the chemical behavior of fluorine, and important additions and improvements have been introduced in the apparatus employed for preparing and experimenting with the gas. M. Moissan now gathers together the results of these subsequent researches - some of which have been published by him from time to time as contributions to various French scientific journals, while others have not hitherto been made known - and publishes them in a long but most interesting paper in the October number of the Annales de Chimîe et de Physique. Inasmuch as the experiments described are of so extraordinary a nature, owing to the intense chemical activity of fluorine, and are so important as filling a long existing vacancy in our chemical literature, readers of Nature will doubtless be interested in a brief account of them.
Improved Apparatus For Preparing Fluorine
In his paper of 1887, the main outlines of which were given in Nature at the time (1887, vol. xxxvii., p. 179), M. Moissan showed that pure hydrofluoric acid readily dissolves the double fluoride of potassium and hydrogen, and that the liquid thus obtained is a good conductor of electricity, rendering electrolysis possible. It will be remembered that, by passing a strong current of electricity through this liquid contained in a platinum apparatus, free gaseous fluorine was obtained at the positive pole and hydrogen at the negative pole. The amount of hydrofluoric acid employed in these earlier experiments was about fifteen grms., about six grms. of hydrogen potassium fluoride, HF.KF, being added in order to render it a conductor. Since the publication of that memoir a much larger apparatus has been constructed, in order to obtain the gas in greater quantity for the study of its reactions, and important additions have been made, by means of which the fluorine is delivered in a pure state, free from admixed vapor of the very volatile hydrofluoric acid. As much as a hundred cubic centimeters of hydrofluoric acid, together with twenty grms. of the dissolved double fluoride, are submitted to electrolysis in this new apparatus, and upward of four liters of pure fluorine is delivered by it per hour.
This improved form of the apparatus is shown in the accompanying figure (Fig. 1), which is reproduced from the memoir of M. Moissan. It consists essentially of two parts - the electrolysis apparatus and the purifying vessels. The electrolysis apparatus, a sectional view of which is given in Fig. 2, is similar in form to that described in the paper of 1887, but much larger.
The U-tube of platinum has a capacity of 160 c.c. It is fitted with two lateral delivery tubes of platinum, as in the earlier form, and with stoppers of fluorspar, F, inserted in cylinders of platinum, p, carrying screw threads, which engage with similar threads upon the interior surfaces of the limbs of the U-tube. A key of brass, E, serves to screw or unscrew the stoppers, and between the flange of each stopper and the top of each branch of the U-tube a ring of lead is compressed, by which means hermetic closing is effected. These fluorspar stoppers, which are covered with a coating of gum lac during the electrolysis, carry the electrode rods, t, which are thus perfectly insulated. M. Moissan now employs electrodes of pure platinum instead of irido-platinum, and the interior end of each is thickened into a club shape in order the longer to withstand corrosion. The apparatus is immersed during the electrolysis in a bath of liquid methyl chloride, maintained in tranquil ebullition at -23°. In order to preserve the methyl chloride as long as possible, the cylinder containing it is placed in an outer glass cylinder containing fragments of calcium chloride; by this means it is surrounded with a layer of dry air, a bad conductor of heat.
The purifying vessels are three in number. The first consists of a platinum spiral worm-tube of about 40 c.c. capacity, immersed also in a bath of liquid methyl chloride, maintained at as low a temperature as possible, about -50°. As hydrofluoric acid boils at 19.5° (Moissan), almost the whole of the vapor of this substance which is carried away in the stream of issuing fluorine is condensed and retained at the bottom of the worm. To remove the last traces of hydrofluoric acid, advantage is taken of the fact that fused sodium fluoride combines with the free acid with great energy to form the double fluoride HF.NaF. Sodium fluoride also possesses the advantage of not attracting moisture. After traversing the worm condenser, therefore, the fluorine is caused to pass through two platinum tubes filled with fragments of fused sodium fluoride, from which it issues in an almost perfect state of purity. The junctions between the various parts of the apparatus are effected by means of screw joints, between the nuts and flanges of which collars of lead are compressed. During the electrolysis these leaden collars become, where exposed to the gaseous fluorine, rapidly converted into lead fluoride, which being greater in bulk causes the joints to become hermetically sealed.
In order to effect the electrolysis, twenty-six to twenty-eight Bunsen elements are employed, arranged in series. An ampere meter and a commutator are introduced between the battery and the electrolysis apparatus; the former affording an excellent indication of the progress of the electrolysis.

FIG. 1. - FLUORINE APPARATUS.
As the U-tube contains far more hydrofluoric acid than can be used in one day, each lateral delivery tube is fitted with a metallic screw stopper, so that the experiments may be discontinued at any time, and the apparatus closed. The whole electrolysis vessel is then placed under a glass bell jar containing dry air, and kept in a refrigerator until again required for use. In this way it may be preserved full of acid for several weeks, ready at any time for the preparation of the gas. Considerable care requires to be exercised not to admit the vapor of methyl chloride into the U-tube, as otherwise violent detonations are liable to occur. When the liquid methyl chloride is being introduced into the cylinder, the whole apparatus becomes surrounded with an atmosphere of its vapor, and as the platinum U-tube is at the same instant suddenly cooled the vapor is liable to enter by the abducting tubes. Consequently, as soon as the current is allowed to pass and fluorine is liberated within the U-tube, an explosion occurs. Fluorine instantly decomposes methyl chloride, with production of flame and formation of fluorides of hydrogen and carbon, liberation of chlorine, and occasionally deposition of carbon.
In order to avoid this unpleasant occurrence, when the methyl chloride is being introduced the ends of the lateral delivery tubes are attached to long lengths of caoutchoue tubing, supplied at their ends with calcium chloride drying tubes, so as to convey dry air from outside the atmosphere of methyl chloride vapor. If great care is taken to obtain the minimum temperature, this difficulty may be even more simply overcome by employing a mixture of well pounded ice and salt instead of methyl chloride; but there is the counterbalancing disadvantage to be considered, that such a cooling bath requires much more frequent renewal.
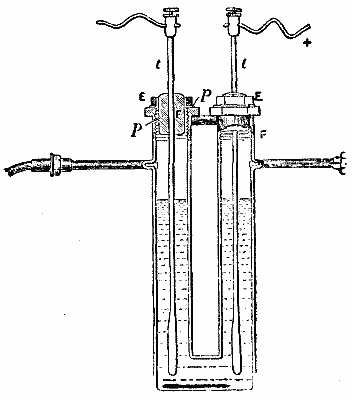
FIG. 2.
Chemical Reactions Occurring During The Electrolysis
In the paper of 1887, M. Moissan adopted the view that the first action of the electric current was to effect the decomposition of the potassium fluoride contained in solution in the hydrofluoric acid, fluorine being liberated at the positive pole and potassium at the negative terminal. This liberated potassium would at once regenerate potassium fluoride in presence of hydrofluoric acid, and liberate its equivalent of hydrogen:
KF = K + F.
K + HF = KF + H.
But when the progress of the electrolysis is carefully followed, by consulting the indications of the amperemeter placed in circuit, it is found to be by no means as regular as the preceding formulae would indicate. With the new apparatus, the decomposition is quite irregular at first, and does not attain regularity until it has been proceeding for upward of two hours. Upon stopping the current and unmounting the apparatus, the platinum rod upon which the fluorine was liberated is found to be largely corroded, and at the bottom of the U-tube a quantity of a black, finely divided substance is observed. This black substance, which was taken at first to be metallic platinum, is a complex compound containing one equivalent of potassium to one equivalent of platinum, together with a considerable proportion of fluorine.
Moreover, the hydrofluoric acid is found to contain a small quantity of platinum fluoride in solution. The electrolytic reaction is probably therefore much more complicated than was at first considered to be the case. The mixture of acid and alkaline fluoride furnishes fluorine at the positive terminal rod, but this intensely active gas, in its nascent state, attacks the platinum and produces platinum tetrafluoride, PtF; this probably unites with the potassium fluoride to form a double salt, possibly 2Kl.PtF, analogous to the well known platinochloride 2KCl.PtCl; and it is only when the liquid contains this double salt that the electrolysis proceeds in a regular manner, yielding free fluorine at the positive pole, and hydrogen and the complex black compound at the negative pole.
Continue to:


