Gum Arabic And Its Modern Substitutes. Part 3
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
Gum Arabic And Its Modern Substitutes. Part 3
Besides obtaining this curve for change in viscosity from maximum amount of metarabin to no metarabin at all, we also traced the decrease in viscosity of the "maximum" solution by dilution with water. The following numbers were thus obtained, and plotted out into a curve.
Having obtained this curve, we are now in a position to follow up the hypothesis by calculating the surplus amount of insoluble matter in a Ghatti. For, let it be conceded that the solution of any Ghatti leaving an insoluble residue is a mixture of arabin and metarabin in the same ratio as our "maximum" solution, only more diluted with water, then from the found viscosity we obtain a point on the curve for dilution, which gives the percentage of dissolved matter.
Now to show the use of this: The Z value for a 10 per cent. solution of the second Ghatti at 15° C. is 2,940. This corresponds on the curve to 8.4 dissolved matter. 10-8.4 = 1.6 grammes in 10 grammes, which is insoluble.
CHANGE OF VISCOSITY WITH DILUTION--"MAXIMUM" SOLUTION. 15° C. TEMPERATURE.
| Percentage. | η | Z. |
| 10 | 0.55250 | 4,456 |
| 9 | 0.42850 | 3,456 |
| 8 | 0.35120 | 2,832 |
| 7 | 0.27660 | 2,230 |
| 6 | 0.22290 | 1,797 |
| 5 | 0.16810 | 1,355 |
| 4 | 0.11842 | 955 |
| 3 | 0.08020 | 647 |
| 2 | 0.06190 | 499 |
| 1 | 0.03610 | 291 |
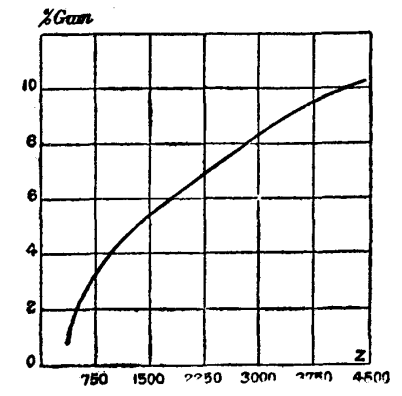
Curve of Variation in Viscosity on Dilution of the "Maximum" Solution.
We have already shown that a "maximum" viscosity solution of this gum is formed when 6.7 per cent, of thin gum arabic is added to it, and therefore 6.7 parts of a thin gum arabic are required to bring 16 parts of metarabin into solution. A convenient rule, therefore, in order to obtain complete solution of a Ghatti gum is to add half the weight in thin gum of the insoluble metarabin found from the viscosity determination. But the portion of the gum which dissolved is made up in a similar manner (being a diluted "maximum" solution).
Therefore the 84 per cent. of soluble matter contains 58 parts of metarabin, and the total metarabin in this gum is 58 + 16 = 74 per cent, on the dry gum.
With these solutions of high viscosity some other work was done which may be of interest. The temperature curves of the mixtures marked E, G, and F were obtained between 60° C. and 15° C. The two former curves showed a direction practically parallel to that at the 10 per cent. solutions, and as they were approaching to the "maximum" solution, this is what one would expect. Mr. S. Skinner, of Cambridge, was also good enough to determine the electrical resistances of these solutions and the Ghattis and gum arabics employed in their preparation. The electrical resistance of these gum solutions steadily diminishes as the temperature increases, and the curve is similar to those obtained for rate of change with temperature. Although the curves run in, roughly, the same direction, there does not appear to be any exact ratio between the viscosities of two gums say at 15° C. and their electrical resistances at the same temperature; hence it would not seem possible to substitute a determination of the electrical resistance for the viscosity determination.
The results appear to be greatly influenced by the amount of mineral matter present, gums with the greatest ash giving lower resistances.
Experiments were conducted with two Ghattis and two gum arabics, besides the mixtures marked E, F, and H. Comparison of the electrical resistances with the viscosities at 15° C. shows the absence of any fixed ratio between them.
| Gum or Mixture. | °C. | Ohms Resistance. | Z Viscosity at 15° C. |
| Ghatti, 1 | 10 | 5,667 | 1,490 |
| Ghatti, 2 | 15 | 2,220 | 2,940 |
| Arabic 1 | 15 | 1,350 | 605 |
| Arabic 2 | 10 | 2,021 | 449 |
| Mixture F | 15 | 1,930 | 787 |
| Mixture E | 11.3 | 2,058 | 3,919 |
While performing these experiments, an attempt was made to obtain an "ash-free" gum, in order to compare its viscosity with that of the same gum in its natural state. A gum low in ash was dissolved in water, and the solution poured on to a dialyzer, and sufficient hydrochloric acid added to convert the salts into chlorides. When the dialyzed gum solution ceased to contain any trace of chlorides, it was made up to a 10 per cent. solution, and its viscosity determined under 100 mm. pressure, giving the following results at 15° C.:
| -------- | η | Z |
| Natural gum | 0.05570 | 449 |
| "Ash-free" gum | 0.05431 | 438 |
Thus showing that the viscosity of pure arabin is almost identical with that of its salts in gum.
The yield of furfuraldehyde by the breaking down of arabin and metarabin was thought possibly to be of some value in differentiating the natural gums from one another, but we have not succeeded in obtaining results of much value. 0.2 gramme of a gum were heated with 100 c.c. of 15 per cent. sulphuric acid for about 2½ hours in an Erlenmeyer flask with a reflux condenser. After this period of time, further treating did not increase the amount of furfuraldehyde produced. The acid liquid, which was generally yellow in color, was then cooled and neutralized with strong caustic soda. The neutral or very faintly alkaline solution was then distilled almost to dryness, when practically the whole of the furfuraldehyde comes over. The color produced by the gum distillate with aniline acetate can now be compared with that obtained from some standard substance treated similarly. The body we have taken as a standard is the distillate from the same weight of cane sugar. The tint obtained with the standard was then compared with that yielded by the gum distillate from which the respective ratios of furfuraldehyde are obtained.
The following table shows some of these results:
| Substance. | Comparative Yield of Furfuraldehyde. | Amount of Glucose Produced. |
| Cane sugar | 1.00 | .. |
| Starch | 0.50 | .. |
| Gum arabic | 1.33 | 34.72 |
| Gum arabic | 1.20 | 43.65 |
| Ghatti, 1 | 1.00 | 26.78 |
| Ghatti, 2 | 1.33 | 22.86 |
| Metarabin | 1.75 | .. |
The amount of reducing sugar calculated as glucose is also appended. This was estimated in the residue left in the flask after distillation by Fehling's solution in the usual way. The yields of furfuraldehyde would appear to have no definite relation to the other chemical data about a gum, such as the potash and baryta absorptions or the sugar produced on inversion.
The action of gum solutions upon polarized light is interesting, especially in view of the fact that arabin is itself strongly laevo-rotatory α = -99°, while certain gums are distinctly dextro-rotatory. Hence it is evident that some other body besides arabin is present in the gum. We have determined the rotatory power of a number of gum solutions, the results of which are subjoined. On first commencing the experiments we experienced great difficulty from the nature of the solutions. Most of them are distinctly yellow in color and almost opaque to light, even in dilute solutions such as 5 percent. We found it necessary first to bleach the gums by a special process; 5 grammes of gum are dissolved in about 40 c.c. of lukewarm water, then a drop of potassium permanganate is added, and the solution is heated on a water bath with constant stirring until the permanganate is decomposed and the solution becomes brown. A drop of sodium hydrogen sulphate is now added to destroy excess of permanganate.
At the same time the solution becomes perfectly colorless.
It can now be cooled down and made up to 100 c.c., yielding a 5 per cent. solution of which the rotatory power can be taken with ease. Using a 20 mm. tube and white light the above numbers were obtained.
| Gum or Dextrin. | Solution used. Per Cent. | α |
| Aden, 1 | 5 | - 33.8 |
| Cape, 2 | 5 | + 28.6 |
| Indian, 3 | 5 | + 66.2 |
| Eastern, 4 | 5 | - 26.0 |
| Eastern, 5 | 5 | - 30.6 |
| Senegal, 6 | 5 | - 17.6 |
| Senegal, 7 | 5 | - 18.4 |
| Senegal, 8 | 2½ | - 19.6 |
| Senegal, 9 | 5 | - 38.2 |
| Senegal, 10 | 5 | - 25.8 |
| Amrad | 2½ | + 57.6 |
| Australian, 1 | 5 | - 28.2 |
| Australian, 2 | 5 | - 26.4 |
| Brazilian, 1 | 2½ | - 36.8 |
| Brazilian, 2 | 2½ | + 21.0 |
| Dextrin, 1 | 5 | +148.0 |
| Dextrin, 2 | 5 | +133.2 |
| Ghatti, 1 | 5 | - 39.2 |
| Ghatti, 2 | 5 | - 80.4 |
These numbers do not show any marked connection between the viscosity, etc., of a gum and its specific rotatory power.
When gum arabic solution is treated with alcohol the gum is precipitated entirely if a large excess of spirit be used. With a view to seeing if the precipitate yielded by the partial precipitation of a gum solution was identical in properties to the original gum, we examined several such precipitates from various gums to ascertain their rotatory power. We found in each case that the specific rotatory power of the alcohol precipitate redissolved in water was not the same as that of the original gum. In other words these gums contained at least two bodies of different rotatory powers, of which one is more soluble in alcohol than the other. O'Sullivan obtained similar results with pure arabin. The experiments were conducted in the following manner:
(a.) Five grammes of a dextro-rotatory gum (No. 3 in table) were dissolved in 20 c.c. of water. To the solution was added 90 c.c. of 95 per cent. alcohol. The white precipitate which formed was thrown on to a tared filter and washed with 30 c.c. more alcohol. The total filtrate therefore was 140 c.c. The precipitate was dried and weighed = 2.794 grammes or 55.88 per cent. of the total gum. The precipitate was then redissolved in water, bleached as before and diluted to a 5 per cent. solution. This was then examined in the polarimeter. Readings gave the value α = +58.4°. The previous rotatory power of the gum was +66°. Now the alcohol was driven off from the filtrate, which, allowing for the 11.95 per cent. of water in the gum, should contain 32.17 per cent. of gum. The alcohol-free liquid was then diluted to a known volume (for 5 per cent, solution), and α found to be + 57.7°. This experiment was then repeated again, using 5 grammes of No. 3, when 3.5805 grammes of precipitate were obtained, using the same volumes of alcohol and water.
The precipitate gave α = +57.4°; the filtrate treated as before, only the percentage of gum dissolved being directly determined instead of being calculated by difference, gave α = + 52.5°.
(b.) Another gum (No. 9) with α = -38.2° and containing 13.86 per cent, of moisture, gave 2.3315 grms. of precipitate when similarly treated. The precipitate gave when redissolved in water α = -20.8°. The filtrate containing 39.5 per cent, real gum gave α = -67.5°, so that the least laevo-rotatory gum. was precipitated by the alcohol.
The Ghattis apparently are all laevo-rotatory, and give much less alcoholic precipitates than the gum arabic. The precipitation moreover was in the opposite direction, that is, the most laevo-rotatory gum was thrown down by the alcohol. The appended table shows the nature of the precipitates and the respective amounts from two Ghattis and two gum arabics. It will be observed that the angle of rotation in three of the cases is decidedly less both for precipitate and filtrate than for the original solution:
SPECIFIC ROTATORY POWERS OF GUMS.
| Gum used. | Weight Gum Waken. Grms. | Weight Alcohol Precipitate. | Weight Gum Filtrate. | α Original Gum. | α Alcohol Precipitate. | α Filtrate. | |
| 3 | a b | 5 5 | 2.7940 3.5805 | 1.9415 0.8910 | + 66.2 | + 58.4 + 57.4 | + 53.7 - 52.5 |
| 9 | a b | 5 4.9620 | 2.3315 2.3310 | 2.3736 2.4180 | - 38.2 | - 20.8 - 19.4 | - 67.5 - 63.4 |
| Ghatti: | a b | 3.4900 3.2450 | 0.3925 0.4605 | 2.7920 2.8385 | -140.8 | -104.2 -106.0 | - 76.0 - 72.4 |
| Ghatti | a b | 2.2550 2.6635 | 0.2900 0.2845 | 1.8078 2.3360 | -147.05 | -106.04 -102.04 | + 68.0 - 66.2 |
The hygrometric nature of a gum or dextrin is a point of considerable importance when the material is to be used for adhesive purposes. The apparatus which we finally adopted after many trials for testing this property consists simply of a tinplate box about 1 ft. square, with two holes of 2 in. diameter bored in opposite sides. Through these holes is passed a piece of wide glass tubing 18 in. long. This is fitted with India rubber corks at each end, one single and the other double bored. Through the double bored cork goes a glass tube to a Woulffe's bottle containing warm water. A thermometer is passed into the interior of the tube by the second hole. The other stopper is connected by glass tubing to a pump, and thus draws warm air laden with moisture through the tube. Papers gummed with the gums or dextrins, etc., to be tested are placed in the tube and the warm moist air passed over them for varying periods, and their proneness to become sticky noted from time to time. By this means the gums can be classified in the order in which they succumbed to the combined influences of heat and moisture. We find that in resisting such influences any natural gum is better than a dextrin or a gum substitute containing dextrin or gelatin.
Continue to:


