Chapter II. Sugar Cookery Classification Of The Carbohydrates
Description
This section is from the book "Experimental Cookery From The Chemical And Physical Standpoint", by Belle Lowe. Also available from Amazon: Experimental cookery.
Chapter II. Sugar Cookery Classification Of The Carbohydrates
The carbohydrates are divided into three groups: the monosaccharids, disaccharids, and polysaccharids. The monosaccharids are composed of one saccharid or sugar group. They are sometimes called simple sugars. The monosaccharids differ from each other in the number of carbon groups and in the molecular arrangement. The monosaccharids contain alcohol groups (HCOH), the number of which may vary from one to six. In addition to the alcohol group a monosaccharid contains either an aldehyde (HC = O) or a ketone (C = O) group. Thus they are aldehyde or ketone derivatives of complex alcohols and as such are called aldoses or ketoses.
A biose is a sugar with two carbon groups, an alcohol group being attached to one carbon and an aldehyde group to the other. A triose has three carbon groups and has alcohol groups attached to two carbons and either an aldehyde or ketone to the third carbon. The common monosac-charids in foods are the hexoses, which contain six carbon groups. Five of the carbons have alcohol groups, but the sixth has an aldehyde or ketone. The common hexoses are dextrose and levulose, the former being an aldose, and the latter a ketose. The following formulas though not conveying the exact arrangement of the molecule illustrate the above points.
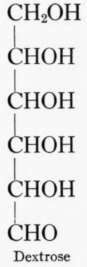
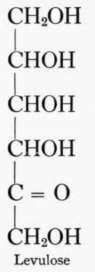
All sugars higher than tetroses may assume two structural forms, the pyran and the furan. The pyran form for a hexose sugar is a ring composed of five carbons and one oxygen with one carbon outside the ring; the furan form is a ring of four carbons and one oxygen with two carbons outside the ring.

Pyran

Beta-fructopyranose
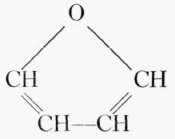
Furan

Beta-fructofuranose
The two structural forms of the sugars are represented by the terms glucopyranose and glucofuranose for glucose, fructopyranose and fructo-furanose for fructose, etc. To avoid confusion of nomenclature, prefixes are added to the terms indicating structural form to differentiate the stereochemical forms (the same chemical and basic structural form but with different spacial arrangements of the hydrogen and hydroxyl groups). Thus the prefixes alpha-, beta-, gamma-, etc., added to the name of the sugar with the ending denoting the structural form, such as alpha-gluco-pyranose, beta-glucofuranose, definitely identify each sugar.
Haworth and his coworkers at Birmingham have worked out in detail the way in which sugar units are united into chains. Haworth states that there are several ways in which two glucose units may be joined through the intermediary of a common oxygen atom. But of the several OH positions available for providing the union of two glucose units, it has been found that the union is formed through the first carbon of one unit and the fourth carbon of the other glucose unit. Maltose is formed, as shown, by the union of two alpha-glucopyranose units. The union of about 30 alpha-glucopyranose units in this manner, according to Haworth forms starch, though all investigators do not agree on the number of glucose units in the starch chain. (See discussion of starch in Chapter XI (Flour And Bread).)
Cellulose is composed of many beta-cellobiose units. In a manner similar to that in which maltose is formed, two beta-glucopyranose units form a-maltose beta-cellobiose. Sucrose is composed of one alpha-glucopyranose united through the first carbon to beta-fructopyranose.
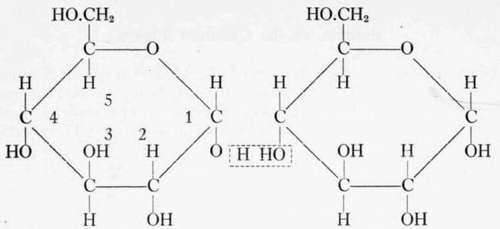
The union of two a - glucopyranose units gives a - maltose.

The disaccharids contain two monosaccharid sugar groups; the poly-saccharids contain many sugar groups. There are also trisaccharids. There is always the possibility that the trisaccharids and some of the other rare sugars may become commercially important, but at the present time they are little used except in scientific work.
The common disaccharids found in foods are sucrose, maltose, and lactose. Sucrose is composed of one dextrose and one levulose group; maltose is made up of two dextrose groups; and lactose is composed of one dextrose and one galactose group.
Following is a list of the common carbohydrates used in foods. For a complete list a physiological or organic chemistry may be consulted.
Monosaccharids :
Dextrose or glucose Levulose or fructose
Disaccharids:
Sucrose Maltose Lactose
Polysaccharids:
Dextrins
Starch
Cellulose
Pectins
Gums
Continue to:


