The Plant Acids. Continued
Description
This section is from the book "Experimental Cookery From The Chemical And Physical Standpoint", by Belle Lowe. Also available from Amazon: Experimental cookery.
The Plant Acids. Continued
Mineral salts of organic acids may be formed during the cooking of fruits in metal containers. Very unpalatable flavors are developed in this way, and the color of the cooked product is darkened. This brings to memory a sample of plum butter sent to the Department. It was nearly charcoal black in color, and as for flavor might have been made from any fruit, for there was little of the original fruit flavor left. The sender stated it had been cooked on the back of the stove in a tin wash boiler for three days and was much perturbed because a neighbor had told her it was poisonous on account of the cooking utensil used. Hence a sample was sent with the request to try it and see if it was poisonous.
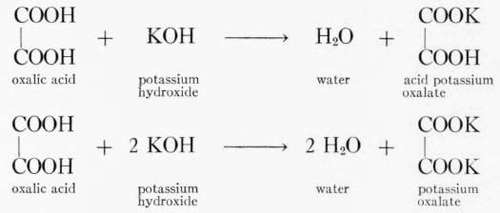
Just as inorganic acids may combine with metals to produce acid or neutral salts according to whether part or all of the hydrogen is replaced by the metal, so in a similar way organic acids may combine with metals forming salts.
Thus oxalic acid gives
Zinc utensils. Some of the metal salts formed with organic acids are injurious when taken into the body. The zinc salts of organic acids have been regarded as toxic. They are formed when foods containing acids are placed in galvanized containers. Galvanized iron contains some zinc, and when fruits, beverages, or even milk are placed in utensils made of it, the acids of the food combine with the zinc, forming salts.
Sale and Badger have reported that zinc is dissolved from galvanized utensils, and the greater the acidity of the food and the longer the food stays in such containers the more zinc dissolved. Even fresh milk contains enough acid to dissolve appreciable amounts of zinc.
Burke, Woodson, and Heller, after investigating the toxicity of buttermilk held in galvanized containers, question the attributing of the toxicity to zinc in many previous investigations, and think that in some cases the toxicity may have been due to the surface of the galvanized container being contaminated with some other substance, possibly arsenic, lead, or antimony.
The tin salts have not been found poisonous, but large amounts of them in a food, such as would result when an acid fruit is cooked for a long time in a tin wash boiler, produce a very dark color and a disagreeable metallic flavor.
Iron salts with acids may cause discoloration in some food products.
Continue to:


