Terpinenol-4
Description
This section is from the book "The Volatile Oils Vol1", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
Terpinenol-4
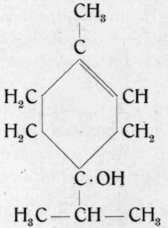
This alcohol which in its general characteristics closely resembles a-terpineol, is derived from terpinene. It has been discovered only recently. In nature it has been found in the oils of juniper, Ceylon cardamoms, nutmeg, marjoram and European wormseed. Synthetically, the active modification has been obtained by shaking sabinene, sabinene hydrate and thujene with dilute sulphuric acid,1) the inactive modification by shaking terpinene di-hydrochloride with dilute potassium hydroxide solution. (Comp. also p. 320.)
1) Wallach, Liebig's Annalen 356 (1907), 218. 2) Ibidem 362 (1908), 280.
The properties of the active modification have been recorded as follows:
B. p. 209 to 212°; d19o 0,9265; aD + 25°4'; nD19o1,4785.2)
Those of the inactive modification are:
B. p. 212 to 214°; d 0,9290; nD 1,4803.3)
This alcohol is known only in the liquid condition. Its odor is less pleasant than that of terpineol. When acted upon in glacial acetic acid solution with hydrohalogen the corresponding terpinene dihydrohalides result. When shaken with dilute sulphuric acid terpinene terpin, m. p. 137°, results. In as much as this hydration is much slower than that of terpineol to terpin hydrate, this serves as a means for the separation of the two alcohols.
Upon oxidation with dilute permanganate solution, v1ter-pinenol-4 yields in the main 1,2,4-trihydroxyterpane, C10H17(OH)3. With its water of crystallization it melts at 116 to 117°, freed therefrom at 128 to 129°; [a]D abt. 21,5°. When distilled with hydrochloric acid, this compound yields carvenone (m. p. of the semicarbazone 200 to 201°), with little cymene. Upon further oxidation with alkaline permanganate solution a mixture of active and inactive a,a'-dihydroxy-a-methyl-a'-Isopropyl-adi-pinic acid, C10H18O6, results. It melts at 205 to 206°, respectively at 188 to 189°. Distilled with steam they readily pass over in the form of the volatile dilactones which melt at 63 to 64° and 72 to 73° respectively. By more energetic oxidation the dihydroxy acid yields w-dimethyl acetonyl acetone as "Abbau" product. The melting point of its dioxime is 137°, that of its semicarbazone 201 to 202°.
1) Wallach, Liebig's Annalen 356 (1907), 215; 360 (1908), 94, 97; 362 (1908), 279; Bed. Berichte 40 (1907), 594.
2) Wallach, Liebig's Annalen 356 (1907), 215.'
3) Ibidem 350 (1906), 155.
Continue to:


