251. Japanese Staranise Oil
Description
This section is from the book "The Volatile Oils Vol2", by E. Gildemeister. Also available from Amazon: The Volatile Oils.
251. Japanese Staranise Oil
Oil of the Fruits
The poisonous fruits of the Japanese staranise tree Illicium religiosum, Sieb. contain 1 p.c. (with reference to the dry fruit4)) of a volatile oil. Its odor is very disagreeable") and has nothing in common with that of the true staranise. d15o 0,984°) to 0,9857); aD -0°507) to -4°5'4); A.V. 1,87); E.V. 12,97); soluble in 5 to 6 vol. of 80 p. c. alcohol with the separation of paraffin7).
1) Chemist and Druggist 53 (1898), 875.
2) Ibidem 54 (1899), 323.
3) Foot note 3 on p. 386.
4) Report of Schimmel & Co. October 1893, Table, p. 42.
5) Schimmel's Bericht September 1885, 29.
6) Report of Schimmel & Co. October 1893, 40. 7) Ibidem April 1909, 52.
Tardy1) extracted dried and pulverized fruits with petroleum ether and obtained 0,4 p.c. of oil after evaporation of the solvent. The oil contained eugenol which was changed to vanillin. From the lower fractions of that portion of the oil which had not been taken up by caustic alkali solutions, Tardy obtained two terpene chlorhydrates. Fraction 170 to 177° consisted principally of cineol of which the bromide was prepared and from which the cineol was regenerated. Fraction 220 to 230° yielded upon oxidation anisic acid, thus revealing the presence of anethol or methylchavicol. Oxidation of fraction 230 to 235° yielded piper-onylic acid melting at 228°, thus indicating the presence of safrol. The higher fractions contained mainly sesquiterpenes. The distillation residue, which was considerable, yielded upon saponification palmitic acid melting at 62°.
An oil examined by Schimmel & Co.2) congealed at - 18° with separation of safrol. It also contained cineol (resorcinol compound). A higher fraction presumably contained linalool. Anethol, however, seemed to be wanting almost entirely.
Oil of the Leaves
The oil from the leaves of the Japanese staranise tree has been examined by J. F. Eykman3). Upon distillation of the leaves he obtained 0,44 p.c. of volatile oil with a specific gravity of 1,006 at 16,5° and a specific angle of rotation [a]D - 8° 6'. It contained a terpene boiling between 173 and 176° (d 0,855; aD - 22,5°) and 25 p.c. of liquid anethol (methyl chavicol?), which yielded nitroanisic acid melting at 174°.
According to a second communication by the same author4), the oil from the leaves and the unripe fruits contains eugenol, a terpene boiling between 168 and 172° named "shikimene" (d 0,865) and "shikimol" or safrol.
1) E. J. Tardy, Etude analytique sur quelques essences du genre anisique. These, Paris 1902, p. 42.
2) Report of Schimmel & Co. April 1909, 57.
3) Mitteilungen d. deutsch. Gesellsch. fur Natur- u. Volkerkunde Ost-asiens 23 (1881); Berl. Berichte 14 (1881), 1720.
4) Recueil trav. chim. des P.-B 4 (1885), 32; Berl. Berichte 18 (1885), 281. Abstracts.
It is noteworthy that in connection with this shikimol Eykman determined the constitution of safrol.
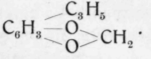
Continue to:


