Methylal. Methylenedimethyl Ether. CH2(OCH3)2. Not officinal
Description
This section is from the book "A Text-Book Of Pharmacology, Therapeutics And Materia Medica", by T. Lauder Brunton. Also available from Amazon: A text-book of pharmacology, therapeutics and materia medica.
Methylal. Methylenedimethyl Ether. CH2(OCH3)2. Not officinal
Characters. - A mobile, colourless, volatile liquid, boiling at
42° C.; sp. gr. 0.8551. Odour like chloroform and acetic ether, with a burning aromatie taste.
Preparation. - By distilling methyl alcohol with an oxidising mixture of dioxide of manganese and sulphuric acid, and adding potash to the distillate to separate methyl formate.
Dose. - 1 gramme (15 gr.).
Action and Use. - It is a local anaesthetic. It produces in dogs anaesthesia, followed by deep sleep. It is rapidly eliminated, and is said not to produce any bad after-effects. It is recommended as a local anaesthetic to the skin, and as an analgesic to the stomach; but as yet it has not been much employed.
Urethane. Ethyl Carbamate. Not officinal.
Urethane is a general term for the ethereal salts of carbamic acid; but ethyl carbamate is the most important of them, so it is usually called par excellence urethane, just as ethylic ether is usually called simply ether.
Carbamic acid, CO.NH2.OH, or H, is not known i the free state. Its ammonium salt forms an important constituent of the officinal ammonium carbonate. The general formula for the salts of carbamic acid is and of ethyl carbamate, (urethane) Ammonium carbamate is and its relationship, as well as that of urethanes, to urea is seen by referring to its rational formula, (see p. 636).


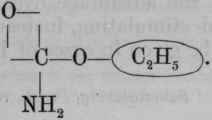
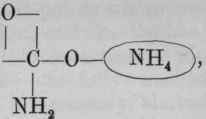
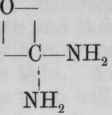
Preparation. - From commercial ethyl chlorocarbonate, by adding solution of ammonia, which converts it into ethyl carbamate (urethane). This is removed by ether, which, with the water, is then distilled off. The urethane which remains is purified by distilling, and then dried over sulphuric acid.
Dose. - 4-8 gr., repeated; or 15-30 gr., or more, in one dose.
Action. - The value of this drug and, though to a less extent, of other urethanes as a hypnotic was discovered by Schmiedeberg, from the consideration that the alcohol radical in it ought to exert a sedative or paralysing action on the cerebrum (p. 764); while the amidogen in it ought to have a somewhat stimulating action on the medulla and cord (p. 602). It ought, therefore, to have a soporific action, like chloral, and yet be free from the danger of paralysing the respiratory centre or heart.
In frogs, doses of 20 to 30 milligrammes cause a condition in which the animals are very readily hypnotised without affecting the respiration or co-ordination of movement. Larger doses diminish voluntary motion without affecting reflex excitability, which is, however, paralysed by still larger doses.
In warm-blooded animals, the same symptoms are produced; and may be ascribed to a blunting of the functions of the cerebral hemispheres, diminution of voluntary motion, and of the perception of sensory stimuli, ending in deep narcosis. In dogs, urethane causes a staggering gait, and, in large doses, vomiting. Urethane stimulates the respiration and, unlike chloral, does not diminish the blood-pressure or affect the heart.1
Uses. - Urethane is a pure hypnotic, and may be used instead of bromide of potassium and chloral in those cases in which, from overwork, worry or other cause, there is an inability to sleep (p. 199). Urethane produces the necessary tranquillity conducive to normal sleep. It is best given in small doses frequently repeated (4 gr.), as vomiting may occur from large doses. It has the advantage over chloral of not affecting the circulation and stimulating, instead of depressing, the respiration. It may be given in cases of heartrdisease and of Bright's disease. When the tension is high, however, it may be less active than chloral, as it does not lessen the tension like chloral, and thus does not reduce the flow of blood through the brain. It is an antidote to strychnine.
1 Schmiedeberg, Pract., vol. xxxv., p. 275.
Continue to:


