3. Butter
Description
This section is from the book "Practical Dietetics With Special Reference To Diet In Disease", by William Gilman Thompson. Also available from Amazon: Practical Dietetics with Special Reference to Diet in Disease.
3. Butter
Butter is made from cream by the mechanical rupture of the albuminous envelopes which inclose the fat globules. The globules then adhere together in small masses. The rupture is accomplished by churning, and after the butter is of sufficient density, salt in small proportion is added, and the butter is "worked" over with a spatula and frequently washed.
An average sample of good butter contains: Water, 11.83 per cent; fat, 82.76 per cent; casein, 0.18 per cent; milk salt, 5.22 per cent (König). The fats comprise six neutral varieties, four of which, being volatile, give to it taste and odour (Chevreu).
The quantity of casein in butter can be roughly estimated by melting a specimen in a test tube, when the casein forms a layer at the bottom.
Butter should not have less than 0.5 to 2 per cent nor more than 8 per cent of salt added.
Perfectly fresh butter made without salt is comparatively tasteless and keeps poorly. The decomposition of its nitrogenous material liberates a ferment which separates fatty acids and glycerin.
Butter is adulterated by dealers by beating it up with water and by adding other fats, especially suet and oleomargarine. The sophistication with water is easily detected by melting, when the sample separates into two layers.
Fresh butter if kept too long, especially in hot weather, soon becomes rancid, bitter, and unwholesome.
Butter which contains too much water and too many ingredients other than the fat has not been sufficiently " worked," and is also likely to become rancid and cause violent gastric derangement. Very thorough washing of butter when first made will remove much of its proteid substance and tend to prevent its fermentation; and fresh butter which has once become rancid may be rendered less so by melting and shaking it in boiling water which is several times renewed and then pouring it into ice-cold water. It is then called "smalt." The addition of salt to butter in the strength of twenty to twenty-five grammes per kilogramme postpones or prevents the fermentation, and sugars or sirups in which the butter may be immersed have the same effect. So also does protecting it from atmospheric air in hermetically sealed jars set in cold water, or simply keeping it under fresh water which is renewed two or three times a day. Sometimes weak solutions of tartaric or acetic acid, three grammes to the litre (Hayem), are employed for this purpose.
Pure fresh-butter fat is not crystalline, but old or rancid butter fat becomes so.
Strong-tasting butter is not necessarily unwholesome, for the flavour may have been derived from the cow having fed upon dead leaves or turnips, and butter is even more sensitive than milk in transmitting unpleasant flavours.
Butter will not support life for any length of time when taken alone, although 100 grains furnish 837 calories. Carnivora fed upon an exclusive diet of meat and fat do not grow fat, therefore the original Banting treatment for obesity (see Obesity) has been modified by Ebstein and others by the free addition of butter to the diet. Taken in connection with other foods, however, butter is a highly digestible and nutritious food, and is often fattening, and it has been wittily said that "if bread is the staff of life, butter is its golden head".
In weak stomachs the digestion of butter is improved by spreading it thoroughly in thin layers upon bread, or allowing it to soak into hot toast. It is thus prevented from floating about in the manner which coats the food and walls of the stomach and retards gastric secretion (Brunton). Most persons eat about an ounce a day of butter, but many eat double that quantity. Its annual production in the United States exceeds one billion pounds.
The use of oleomargarine and butterine as substitutes for butter are described under the heading Fats and Oils.
PLATE II.

Butter x 40.
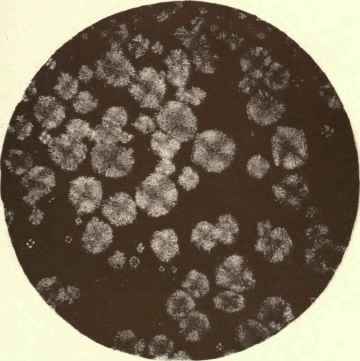
Butterine X 40 Animal Fats, Magnified.
(From Bulletin No. 13, Division of Chemistry, United States Agricultural Bureau).
PHOTO BY CLIFFORD RICHARDSON.
Continue to:


