Chemical Nature And Physical Properties Of Proteins In General
Description
This section is from the book "Chemistry Of Food And Nutrition", by Henry C. Sherman. Also available from Amazon: Chemistry of food and nutrition.
Chemical Nature And Physical Properties Of Proteins In General
Generally speaking, the proteins of different kinds of tissue, and even of the corresponding tissues of different species, are not identical substances. The total number of different proteins occurring in nature must therefore be very great. Of these, some fifty or sixty have been sufficiently isolated and studied to warrant description as chemical individuals. All of these have proven to be very complex substances and in no case has the chemical structure of a natural protein been fully determined. It has, however, been shown that the typical proteins are essentially anhydrides of the following amino acids:
Amino Acids Of Common Proteins
Monaminomonocarboxylic acids Glycine, amino-acetic acid, CH2(NH2) • COOH. Alanine, α-amino-propionic acid, CH3CH(NH2) • COOH. Valine, α-amino-isovaleric acid, (CH3)2CH • CH(NH2) • COOH. Leucine, α-amino-isocaproic acid (α-amino-isobutyl-acetic acid),
(CH3)2CH • CH2 • CH(NH2) • COOH. Phenylalanine, phenyl-α-amino-propionic acid,
C6H5CH2 • CH(NH2) •COOH. Tyrosine, oxyphenyl α-amino propionic acid,
C6H4(OH) • CH2 • CH(NH2) • COOH. Serine, α-amino-ß-hydroxy-propionic acid,
CH2(OH): CH(NH2) • COOH. Cystine (dicysteine), or di-(α-amino-ß-thio-lactic acid),

Monaminodicarboxylic acids Aspartic add, amino-succinic acid,
COOH • CH2 • CH(NH2) • COOH. Glutamic (glutaminic) acid, amino-glutaric acid, COOH • CH2 • CH2 • CH(NH2) • COOH. Diaminomonocarboxylic acids Ornithine, α, δ, diamino-valeric acid,
CH2(NH2) • CH2 • CH2 • CH(NH2) • COOH. Arginine, δ-guanidino-α-amino-valeric acid,
NH (H2N)C • NH • CH2 • CH2 • CH2 • CH(NH2) • COOH.
Lysine,α є, diamino-n-caproic acid,
CH2(NH2) • CH2 • CH2 • CH2 • CH(NH2) • COOH.
Heterocyclic Amino Acids
Histidine, α-amino-ß-imidazol propionic acid.
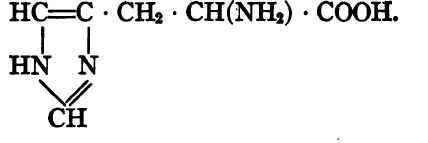
Proline, pyrrolidin-carboxylic acid.
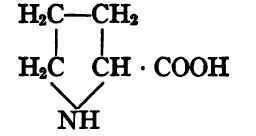
Tryptophane, α-amino-ß-indol-propionic acid.
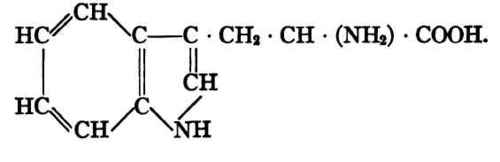
It will be noted that these constituents of the protein molecule differ much in structure among themselves. They are, however, all α-amino acids, i.e., the amino group (or one of them if there be more than one) is attached to the carbon atom adjacent to the carboxyl.
In view of the wide occurrence of the alanine radicle in proteins and the frequency with which we shall have occasion to discuss the behavior of alanine (as a typical amino acid) in metabolism, it may be of interest to point out that several of the amino acids, even including some of unique constitution, may be regarded as derived from alanine by the substitution of a simple or complex radicle for one of the hydrogens on the ß carbon of alanine. Thus by the substitution of an -OH or -SH group one obtains serine or cysteine respectively; by substituting the phenyl or oxyphenyl group, there results phenylalanine or tyrosine; by the imidazole (C3H3N2), histidine; by the indol (C8H6N) radicle, tryptophane.

Alanine.
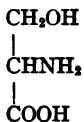
Serine.
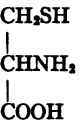
Cysteine.
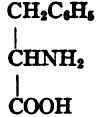
Phenylalanine.
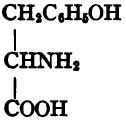
Tyrosine.

Histidine.

Tryptophane.
The linkage of the amino acid radicles in the protein molecule is chiefly through the carboxyl group of one amino acid reacting with the amino group of another. Thus two molecules of glycine combined by elimination of one molecule of water yield glycyl-glycine, which is the simplest of an immense group of anhydrides of amino acids, all of which are called "peptids." Dipeptids contain two amino acid radicles, tripeptids contain three, etc. Fischer, by uniting 7 to 19 amino-acid radicles, has produced synthetic polypeptids which in some of their properties resemble the peptones, the simplest substances usually regarded as true proteins. Peptones are formed in nature by the digestive hydrolysis of ordinary proteins, whose structure is doubtless considerably more complex.
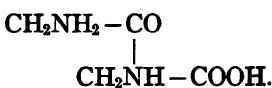
A certain analogy between carbohydrates and proteins may be noted. As starch on hydrolysis yields the polysaccharide dextrins, the disaccharide maltose, and finally as end product the monosaccharide glucose, so the native protein is hydrolyzed through peptones, polypeptids, and di- or tri-peptids, to amino acids. Thus the amino acid bears the same general relation to the protein which glucose bears to starch; and just as the molecular weight of starch is very high and a single starch molecule yields a large (unknown) number of monosaccharide molecules, so the molecular weight of the protein is very high and the protein molecule yields a large (unknown) number of amino acid molecules. There is, however, this important difference: the molecules of monosaccharide resulting from complete hydrolysis of starch are all alike (glucose), whereas the complete hydrolysis of any typical protein yields several of the above-mentioned amino acids, in the case of most proteins from twelve to twenty.
Continue to:


